AIDS Vaccine Candidate Successfully 'Primes' Immune System
By Madeline McCurry-Schmidt
New research led by scientists at The Scripps Research Institute (TSRI), International AIDS Vaccine Initiative (IAVI) and The Rockefeller University shows in mice that an experimental vaccine candidate designed at TSRI can stimulate the immune system activity necessary to stop HIV infection. The findings could provide key information for the development of an effective AIDS vaccine.
The research, published June 18, 2015 in concurrent studies in the journals Cell and Science, represents a leap forward in the effort to develop a vaccine against HIV, which has so far struggled to elicit antibodies (immune system molecules) that can effectively fight off different strains of the virus.
“The results are pretty spectacular,” said Dennis Burton, chair of the TSRI Department of Immunology and Microbial Science and scientific director of two centers at TSRI, the IAVI Neutralizing Antibody Consortium (NAC) and the National Institutes of Health (NIH) Center for HIV/AIDS Vaccine Immunology and Immunogen Discovery (CHAVI-ID).
The Science study was co-led by Burton, TSRI Professor and IAVI NAC Director of Vaccine Design William Schief, and TSRI Professor David Nemazee. The Cell study was co-led by Schief and Michel Nussenzweig, who is Zanvil A. Cohn and Ralph M. Steinman Professor at The Rockefeller University and a Howard Hughes Medical Institute (HHMI) investigator.
A Huge Challenge
The researchers’ long-term goal is to design a vaccine that prompts the body to produce antibodies that bind to HIV and prevent infection.
While many vaccines for other diseases use a dead or inactive version of the disease-causing microbe itself to trigger antibody production, immunizations with “native” HIV proteins are ineffective in triggering an effective immune response, due to HIV’s ability to evade detection from the immune system and mutate rapidly into new strains.
This challenge has led many researchers to believe that a successful AIDS vaccine will need to consist of a series of related, but slightly different proteins (immunogens) to train the body to produce broadly neutralizing antibodies against HIV—a twist on the traditional “booster” shot, where a person is exposed to the same immunogen multiple times.
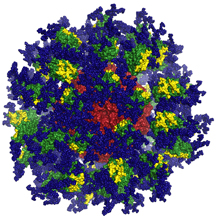
In the new studies, the scientists tested one of these potential proteins, an immunogen called eOD-GT8 60mer, a protein nanoparticle designed to bind and activate B cells needed to fight HIV. The eOD-GT8 60mer was developed in the Schief lab and tested in mouse models engineered by the Nemazee lab to produce antibodies that resemble human antibodies.
Using a technique called B cell sorting, the researchers showed that immunization with eOD-GT8 60mer produced antibody “precursors”—with some of the traits necessary to recognize and block HIV infection. This suggested that eOD-GT8 60mer could be a good candidate to serve as the first in a series of immunizations against HIV.
“The vaccine appears to work well in our mouse model to ‘prime’ the antibody response,” added Nemazee.
In the Cell paper, researchers used the same eOD-GT8 60mer immunogen but used a slightly different mouse model. “The immunogen again launched the immune system in the right direction,” said Schief.
A concurrent study also in Science (led by Professor John Moore of Weill Cornell Medical College and including contributions from Schief, Burton, TSRI Associate Professor Andrew Ward, Ian Wilson, who is Hansen Professor of Structural Biology and chair of the Department of Integrative Structural and Computational Biology at TSRI, and other researchers) showed engineered immunogens also triggered immune responses in rabbit models and non-human primate models.
Next Steps
With eOD-GT8 60mer in the running as a potential contributor to an HIV vaccine, the researchers are now investigating other immunogens that could work in conjunction with it.
Schief said the Nemazee lab’s mouse models will be crucial resources for testing other engineered immunogens. He emphasized the importance of bringing different disciplines together to engineer mouse models, design antibodies and analyze results. “This was a beautiful collaboration of three labs at TSRI,” said Schief.
In addition to Burton, Nemazee and Schief, authors of the Science study, “Priming a broadly neutralizing antibody response to HIV-1 using a germline-targeting immunogen,” include Joseph G. Jardine, Devin Sok, Matthias Pauthner, Daniel W. Kulp, Oleksandr Kalyuzhniy, Bryan Briney, Sergey Menis, Meaghan Jones, Mike Kubitz, Skye Spencer and Yumiko Adachi of TSRI, the IAVI NAC and CHAVI-ID; and Takayuki Ota, Patrick D. Skog, Theresa C. Thinnes and Deepika Bhullar of TSRI. See http://www.sciencemag.org/content/early/2015/06/17/science.aac5894
This study was supported by the IAVI Neutralizing Antibody Consortium and Center, the Ragon Institute of MGH, MIT and Harvard, the Helen Hay Whitney Foundation and the National Institute of Allergy and Infectious Diseases (grants R01-AI073148, P01AI081625) and CHAVI-ID (grant 1UM1AI100663).
In addition to Schief and Nussenzweig, authors of the Cell study, “Immunization for HIV-1 Broadly Neutralizing Antibodies in Human Ig Knock-In Mice,” include Pia Dosenovic, Lotta von Boehmer, Amelia Escolano, Natalia T. Freund, Alexander D. Gitlin, Thiago Oliveira, John Pietzsch, Kai-Hui Yao, and Cassie Liu of The Rockefeller University; Joseph Jardine and Daniel W. Kulp of TSRI, the IAVI NAC and CHAVI-ID; Anna Gazumyan of The Rockefeller University and HHMI; Andrew T. McGuire and Matthew D. Gray of the Fred Hutchinson Cancer Research Center; Louise Scharf of the California Institute of Technology; Albert Cupo and John P. Moore of Weill Medical College; Rogier W. Sanders of Weill Medical College and the University of Amsterdam; Marit J. van Gils of the University of Amsterdam; Michael S. Seaman of Harvard Medical School; Pamela J. Björkman of the HHMI and California Institute of Technology; and Leonidas Stamatatos of the Fred Hutchinson Cancer Research Center and the University of Washington. See http://www.cell.com/cell/abstract/S0092-8674%2815%2900686-8.
This study was supported by the NIH CHAVI-ID (grant UM1 AI100663), the Bill and Melinda Gates Foundation (grants OPP1033115 and 1032144), the NIH (grant AI037526, U19 AI109632, HIVRAD/P01 AI094419-01, HIVRAD/P01 AI082362, HIVRAD/P01 AI100148), the Aids Fonds Netherlands (grant 2012041), an international postdoctoral fellowship from the Swedish Research Council, the NIH National Center for Advancing Translational Sciences (grant UL1 TR000043/KL2TR000151), the NIH Medical Scientist Training Program (grant T32GM07739), a Canadian Institutes of Health Research Fellowship, a Vidi grant from the Netherlands Organization for Scientific Research and a Starting Investigator Grant from the European Research Council.
The studies were published with a companion paper, “HIV-1 Neutralizing Antibodies Induced by Native-like Envelope Trimers,” also in the journal Science.
Send comments to: press[at]scripps.edu