Publications
620. Moore, M. J.; Qin, P.; Yamasaki, N.; Zeng, X.; Keith, D. J.; Jung, S.; Fukazawa, T.; Graham-O'Regan, K.; Wu, Z.-C.; Chatterjee, S.; Boger, D. L. Tetrachlorovancomycin: Total Synthesis of a Designed Glycopeptide Antibiotic of Reduced Synthetic Complexity J. Am. Chem. Soc. 2023, 145, 21132–21141.

619. Moore, M. J.*; Qin, P.*; Keith, D. J.; Wu, Z.-C.; Jung, S.; Chatterjee, S.; Tan, C.; Qu, S.; Cai, Y.; Stanfield, R. L.; Boger, D. L. Divergent Total Synthesis and Characterization of Maxamycins J. Am. Chem. Soc. 2023, 145, 12837–12852. (*Joint cofirst authors)

618. Moore, M. J.; Qin, P.; Keith, D. J.; Boger, D. L. Improved preparative enzymatic glycosylation of vancomycin aglycon and analogues. Tetrahedron 2023, 131, 133211.

617. Wu, Z.-C.; Boger, D. L. 1, 2, 3, 5-Tetrazines: A General Synthesis, Cycloaddition Scope, and Fundamental Reactivity Patterns. J. Org. Chem. 2022, 87,
616. Zhu, Z.; Boger, D. L. Acyclic and Heterocyclic Azadiene Diels–Alder Reactions Promoted by Perfluoroalcohol Solvent Hydrogen Bonding: Comprehensive Examination of Scope. J. Org. Chem. 2022, 87, 14657−14672.
*Highlighted in Youtube Video "Five important papers in organic synthesis (October 2022)"
https://www.youtube.com/watch?v=pJbixeUVE6w&t=318s

615. Rachel M. Gillard, Jiajun Zhang, Richard Steel, Jocelyn Wang, Jessica L. Strull, Bin Cai, Nilanjana Chakraborty, Dale L. Boger* Aryl Annulation: A Powerful Simplifying Retrosynthetic Disconnection. Synthesis 2022, A-P.

614. Ming-Hsiu Yang, Jamie L. Russell, Yuto Mifune, Ying Wang, Hexin Shi, Eva Marie Y. Moresco, Daniel J. Siegwart, Bruce Beutler*, and Dale L. Boger* Next-Generation Diprovocims with Potent Human and Murine TLR1/TLR2 Agonist Activity That Activate the Innate and Adaptive Immune Response. J. Med. Chem. 2022, 65,

613. Wu, Z.-C.; Houk, K.N.; Boger, D.L.*, Svatunek, D.* Mechanistic Insights into the Reaction of Amidines with 1, 2, 3-Triazines and 1, 2, 3, 5-Tetrazines. J. Am. Chem. Soc. 2022, 144,.

612. Zhu, Z.; Boger, D.L. N1/N4 1,4-Cycloaddition of 1,2,4,5-Tetrazines with Enamines Promoted by the Lewis Acid ZnCl2 . J. Org. Chem. 2022, 87, 6288.

611. Zhang, J.*; Paladugu, S.R.*; Gillard, R.M.; Sarkar, A.; Boger, D.L. Tris(4-bromophenyl)aminium Hexachloroantimonate-Mediated Intermolecular C(sp2)-C(sp3) Free Radical Coupling of Vindoline with beta-Ketoesters and Related Compounds. J. Am. Chem. Soc. 2022, 144, 495-502. (*contributed equally)

610. Elwood A Mullins, Jonathan Dorival, Gong-Li Tang, Dale L Boger, Brandt F Eichman*. Structural evolution of a DNA repair self-resistance mechanism targeting genotoxic secondary metabolites. Nat. Commun. 2021, 12, 1.
609. Quiñones, R.E.; Wu, Z.-C.; Boger, D.L. Reaction Scope of Methyl 1,2,3-Triazine-5-carboxylate with Amidines and the Impact of C4/C6 Substitution. J. Org. Chem. 2021, 86 13465-13474.

610. Zeng, X.; Boger, D.L. Total Synthesis of (-)-Strempeliopine. J. Am. Chem. Soc. 2021, 143, 12412-12417.
Highlighted in Synthesis Workshop
(https://www.youtube.com/watch?v=MWXK2elRGOk&t=1s&ab_channel=SynthesisWorkshopVideos)

609. Anindya Sarkar, Anushka C Galasiti Kankanamalage, Qian Zhang, Heng Cheng, Prasanna Sivaprakasam, Joseph Naglich, Chunshan Xie, Sanjeev Gangwar, Dale L Boger. Synthesis, structure-activity relationship studies and evaluation of a TLR 3/8/9 agonist and its analogues. Med. Chem. Res., 2021, 30, 1377.
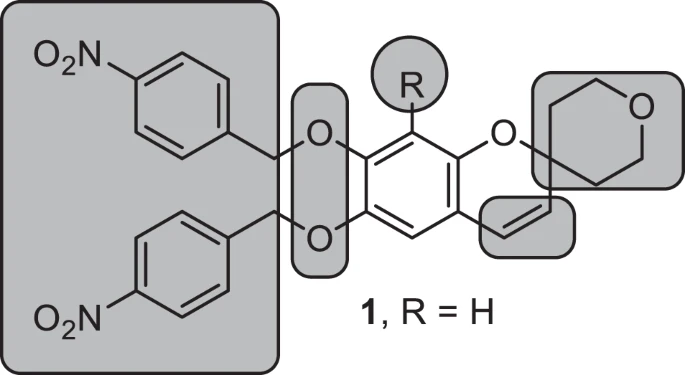
608. Boon, A.B.; Yu, Y.-Y.; Boger, D.L. Total synthesis of (−)-4-desacetoxy-1-oxovindoline: Single atom exchange of an embedded core heteroatom in vindoline. Tetrahedron, 2021, 87, 132217.

607. Litwin, K.; Crowley, V.M.; Suciu, R.M.; Boger, D.L.; Cravatt, B.F. Chemical proteomic identification of functional cysteines with atypical electrohpile reactivities. Tetrahedron Lett., 2021, 67, 152861.

606. Angelbello, A.J.; DeFeo, M.E.; Glinkerman, C.M.; Boger, D.L.; Disney, M.D. Precise targeted cleavage of a r(CUG) repeat expansion in cells by using a small molecule-deglycobleomycin conjugate. ACS Chem. Biol. 2020, 15, 849-855.

605. Zhu, Z.; Glinkerman, C.M.; Boger, D.L. Selective N1/N4 1,4-Cycloaddition of 1,2,4,5-Tetrazines Enabled by Solvent Hydrogen Bonding. J. Am. Chem. Soc. 2020, 142, 20778-20787.

604. Wu, Z.-C.; Cameron, M.D.; Boger, D.L. Vancomycin C-Terminus Guanidine Modifications and Further Insights into an Added Mechanism of Action Imparted by a Peripheral Structural Modification. ACS Infect. Dis. 2020, 6, 2169-2180.

603. Wu, Z.-C.; Boger, D.L. The quest for supernatural products: the impact of total synthesis in complex natural products medicinal chemistry. Nat. Prod. Rep. 2020, 37, 1511-1531.
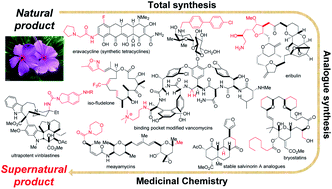
602. Wu, Z.-C.; Boger, D.L. Maxamycins: Durable Antibiotics Derived by Rational Redesign of Vancomycin. Acc. Chem. Res. 2020, 53, 2587-2599.

601. Gartshore, C.; Tadano, S.; Chanda, P.B.; Sarkar, A.; Chowdari, N.S.; Gangwar, S.; Zhang, Q.; Vite, G.D.; Momirov, J.; Boger, D.L. Total Synthesis of Meayamycin and O-Acyl Analogues. Org. Lett. 2020, 22, 8714-8719.

600. Zeng, X; Shukla, V.; Boger, D.L. Divergent Total Syntheses of (-)-Pseudocopsinine and (-)-Minovincinine. J. Org. Chem. 2020, 85, 14817-14826.
Featured Article

599. Moore, M.J.; Qu, S.; Tan, C.; Cai, Y.; Mogi, Y.; Keith, D.J.; Boger, D.L. Next-Generation Total Synthesis of Vancomycin. J. Am. Chem. Soc. 2020, 142, 16039-16050.
Highlighted in C&EN, 2020, 98, 8

598. Kok, B.P.; Ghimire, S.; Kim, W.; Chatterjee, S.; Johns, T.; Kitamura, S.; Eberhardt, J.; Ogasawara, D.; Xu, J.; Sukiasyan, A.; Kim, S.M.; Godio, C.; Bittencourt, J.M.; Cameron, M.; Galmozzi, A.; Forli, S.; Wolan, D.W.; Cravatt, B.F.; Boger, D.L.; Saez, E. Discovery of small-molecule enzyme activators by activity-based protein profiling. Nat. Chem. Biol. 2020, 16, 997-1005
597. Wu, Z.-C.; Isley, N.A.; Okano, A.; Weiss, W.J.; Boger, D.L. C1-CBP-Vancomycin: Impact of a vancomycin C-terminus trimethylammonium cation on pharmacological properties and insights into its newly introduced mechanism of action, J. Org. Chem. 2020, 84, 1365-1375. Invited contribution to SIP entitled “Modern Peptide and Protein Chemistry”.
596. Beekman, A.; Cominetti, M.M.D.; Cartwright, O.C.; Boger, D.L.; Searcey, M. A small molecule drug conjugate (SMDC) of DUPA and a duocarmycin built on the solid phase, Med. Chem. Commun. 2019, 10, 2170–2174.
595. Isley, N.A.; Endo, Y.; Wu, Z.-C.; Covington, B.C.; Bushin, L.B.; Seyedsayamdost, M.R.; Boger, D.L. Total synthesis and stereochemical assignment of streptide, J. Am. Chem Soc. 2019, 141, 17361–17369.

594. Wu, Z.-C.; Boger, D.L. Synthesis, characterization, and cycloaddition reactivity of a monocyclic aromatic 1,2,3,5-tetrazine, J. Am. Chem. Soc. 2019, 141, 16388–16397.

593. Boon, B.A.; Boger, D.L. Triarylamminium radical cation promoted coupling of catharanthine with vindoline: Diastereospecific synthesis of anhydrovinblastine and reaction scope, J. Am. Chem. Soc. 2019, 141, 14349–14355.

592. Plewe, M.B.; Whitby, L.R.; Naik, S.; Brown, E.R.; Sokolova, N.V.; Gantla, V. R.; York, J.; Nunberg, J.H.; Zhang, L.; Kalveram, B.; Freiberg, A.N.; Boger, D.L.; Henkel, G.; McCormack, K. SAR studies of 4-acyl-1,6- dialkylpiperazin-2-one Arenavirus cell entry inhibitors, Bioorg. Med. Chem Lett. 2019, 29, 126620.
591. Zhang, J.; Shukla, V.; Boger, D.L. Inverse Electron Demand Diels-Alder Reactions of Heterocyclic Azadienes, 1-Aza-1,3-Butadienes, Cyclopropenone Ketals, and Related Systems. A Retrospective. J. Org. Chem. 2019, 84, 9397-9445.
590. Radakovic, A.; Boger, D.L. Ultra-potent vinblastine analogues improve on-target activity of the parent microtubulin-targeting compound. Bioorg. Med. Chem. Lett. 2019, 29, 1370–1374.
589. Otrubova, K.; Chatterjee, S.; Ghimire, S.; Cravatt, B.F.; Boger, D.L. N-acyl pyrazoles: Effective and tunable inhibitors of serine hydrolases. Bioorg. Med. Chem. 2019, 27, 1693–1703.
588. Su, L.; Wang, Y.; Wang, J.; Mifune, Y.; Morin, M.D.; Jones, B.T.; Moresco, E.M.Y.; Boger, D.L.; Beutler, B.; Zhang, M.H. Structural Basis of TLR2/TLR1 Activation by the Synthetic Agonist Diprovocim. J. Med. Chem. 2019, 62, 2938-2949.
587. Wu, Z-C.; Boger, D.L. Exploration of the Site-Specific Nature and Generalizability of a Trimethylammonium Salt Modification on Vancomycin: A-ring Derivatives. Tetrahedron. 2019, 75, 3160-3165.
586. Morin, M.D.; Wang, Y.; Jones, B.T.; Mifune, Y.; Su, L.; Shi, H.; Zhang, H.; Moresco, E.M.Y.; Beutler, B.; Boger, D.L. Diprovocims: A New and Exceptionally Potent Class of Toll-like Receptor Agonists. J. Am. Chem. Soc. 2018, 140, 14440-14454.
585. Wu, Z-C.; Isley, N.A.; Boger, D.L. N-Terminus Alkylation of Vancomycin: Ligand Binding Affinity, Antimicrobial Activity, and Site Specific Nature of Quaternary Trimethylammonium Salt Modification. ACS Infect. Dis. 2018, 4, 1468-1474.
584. Wang, Y.; Su, L.; Morin, M.D.; Jones, B.T.; Mifune, Y.; Shi, H.; Wang, K.; Zhan, X.; Liu, A.; Wang, J.; Li, X.; Tang, M.; Ludwig, S.; Hilderbrand, S.; Zhou, K.; Siegwart, D.; Moresco, E.M.Y.; Zhang, H.; Boger, D.L.; Beutler, B. Adjuvant Effect of the Novel TLR1/TLR2 Agonist Diprovocim Synergizes with anti-PD-L1 to Eliminate Melanoma in Mice. Proc. Natl. Acad. Sci. USA. 2018, 115, E8698-E8706.
583. Boyle, K.E.; Boger, D.L.; Wroe, A.; Vasquez, M. Duocarmycin SA, a Potent Antitumor Antiobiotic, Sensitizes Glioblastoma Cells to Proton Radiation. Bioorg. Med. Chem. Lett. 2018, 28, 2688-2692.
582. Glinkerman, C.M.; Boger, D.L. Synthesis, Characterization, and Rapid Cycloadditions of 5-Nitro-1,2,3-triazine. Org. Lett. 2018, 20, 2628-2631.
581. Radakovic, A.; Boger, D.L. High Expression of Class III B-tubulin has no impact on functional cancer cell growth inhibition of a series of Key Vinblastine Analogs. Bioorg. Med. Chem Lett. 2018, 28, 853-865.
580. Okano, A.; Isley, N.A.; Boger, D.L. Total Syntheses of Vancomycin-Related Glycopeptide Antibiotics and Key Analogues. Chem. Rev. 2017, 117, 11952-11993.
579. Boger, D.L. The Difference a Single Atom Can Make: Synthesis and Design at the Chemistry-Biology Interface. J. Org. Chem. 2017, 82, 11961-11980.
578. Lukesh III, J.C.; Carney, D.W.; Dong, H.; Cross, R.M.; Shukla, V.; Duncan, K.K.; Yang, S.; Brody, D.M.; Brutsch, M.M., Radakovic, A.; Boger, D.L. Vinblastine 20' Amides: Synthetic Analogues That Maintain or Improve Potency and Simultaneously Overcome Pgp-Derived Efflux Resistance. J. Med. Chem. 2017, 60, 7591-7604.
577. Quinones, R.E.; Glinkerman, C.M.; Zhu, K.; Boger, D.L. Direct Synthesis of β-Aminoenals through Reaction of 1,2,3-Triazine with Secondary Amines. Org. Lett. 2017, 19, 3568-3571.

576. Okano, A.; Isley, N.A.; Boger, D.L. Peripheral modifications of [Ψ[CH2NH]Tpg4]vancomycin with added synergistic mechanisms of action provide durable and potent antibiotics. Proc. Natl. Acad. Sci. USA, 2017, 114, E5052-E5061.
575. Allemann, O.; Cross, M.; Brutsch, M.M.; Radakovic, A.; Boger, D.L. Key Analogs of a Uniquely Potent Synthetic Vinblastine that contain Modifications fo the C20' Ethyl Substituent. Bioorg. Med. Chem. Lett. 2017, 27, 3055-3059.
574. Kornahrens, A.F.; Cognettalll, A.B.; Brody, D.M.; Matthews, M.L.; Cravatt, B.F.; Boger, D.L. Design of Benzoxathiazin-3-one 1,1-Dioxides as a New Class of Irreversible Serine Hydrolase Inhibitors: Discovery of a Uniquely Selective PNPLA4 Inhibitor. J. Am. Chem Soc. 2017, 139, 7052-7061.
573. Yang, S.L.; Sankar, K.; Skepper, C.K.; Barker, T.J.; Lukesh, J.C.; Brody, D.M.; Brutsch, M.M.; Boger, D.L. Total Synthesis of a Key Series of Vinblastines Modified at C4 that Define the Importance and Surprising Trends in Activity. Chem. Sci. 2017, 8, 1560-1569.
572. Chanda, P. B.; Boyle, K. E.; Brody, D. M.; Shukla, V.; Boger, D. L. Synthesis and Evaluation of Duocarmycin SA Anologs Incorporating the Methyl 1,2,8,8a-Tetrahydrocyclopropa[c]imidazolo[4,5-e]indol-4-one-6-carboxylate (Clml) alkylation subunit. Bioorg. Med. Chem. Lett. 2016, 24, 4779-4786.
571. Glinkerman, C. M.; Boger, D. L. Catalysis of Heterocyclic Azadiene Cycloaddition Reactions by Solvent Hydrogen Bonding: Concise Total Synthesis of Methoxatin. J. Am. Chem. Soc. 2016, 138, 12408-12413.
570. Carney, D. W.; Lukesh, J. C.; Brody, D. M.; Brutsch, M. M.; Boger, D. L. Ultrapotent Vinblastines in which Added Molecular Complexity Further Disrupts the Target Tubulin Dimer-Dimer Interface. Proc. Natl. Acad. Sci. USA. 2016, 113, 9691-9698.
569. Shankar, S.; Whitby, L. R.; Casquilho-Gray, H. E.; et al. Small-Molecule Fusion Inhibitors Bind the pH-Sensing Stable Signal Peptide-GP2 Subunit Interface of the Lassa Virus Envelope Glycoprotein. J. Virol. 2016, 90, 6799-6807.
568. Burman, M. A.; Szolusha, K.; Bind, R.; Kerney, K.; Boger, D. L.; Bilsky, E. J. FAAH Inhibitor OL-135 Disrupts Contextual, but not Auditory, Fear Conditioning in Rats. Behav. Brain Res. 2016, 308, 1-5
567. Allemann, O.; Brutsch, M.; Lukesh, J. C.; Brody, D. M.; Boger, D. L. Synthesis of a Potent Vinblastine: Rationally Designed Added Benign Complexity. J. Am. Chem. Soc. 2016, 138, 8376-8379
566. Morin, M. D.; Wang, Y.; Jones, B. T.; Su, L. J.; Sarakattula, M.M.R.P; Berger, M.; Huang, H.; Beutler, E.K.; Zhang, H.; Beutler, B.; Boger, D. L. Discovery and Structure-Activity Relationships of the Neoseptins: A New Class of Toll-like Receptor-4 (TLR4) Agonists. J. Med. Chem. 2016, 59, 4812-4830
565. Wang, Y.; Su, L.; Morin, M. D.; Jones, B. T.; Whitby, L. R.; Murali, M. R. P.; Huang, H.; Shi, H.; Choi, J. H.; Wang, K.; Moresco, E. M. Y.; Berger, M.; Zhan, X.; Zhang, H.; Boger, D. L.; Beutler, B. TLR4/MD-2 activation by a synthetic agonist with no similarity to LPS. Proc. Natl. Acad. Sci. USA. 2016, 113, E884-E893
564. Sears, J. E.; Boger, D. L. Tandem Intramolecular Diels-Alder/1,3-Dipolar Cycloaddition Cascade of 1,3,4-Oxadiazoles: Initial Scope and Applications. Acc. Chem. Res. 2016, 49, 241-251
563. Janssen, F. J.; Baggelaar, M. P.; Hummel, J. J. A.; Overkleeft, H. S.; Cravatt, B. F.; Boger, D. L.; van der Stelt, M. Comprehensive Analysis of Structure–Activity Relationships of α-Ketoheterocycles as sn-1-Diacylglycerol Lipase α Inhibitors. J. Med. Chem. 2015, 58, 9742-9753.
562. Sears, J. E.; Barker, T. J.; Boger, D. L. Total Synthesis of (-)-Vindoline and (+)-4-epi-Vindoline Based on a 1,3,4-Oxadiazole Tandem Intramolecular [4+2]/[3+2] Cycloaddition Cascade Initiated by an Allene Dienophile. Org. Lett. 2015, 17, 5460-5463.
561. Boger, D. L. Recent Advances in Medicinal Chemistry and Chemical Biology. Bioorg. Med. Chem. Lett. 2015, 25, 4713-4713.
560. Boger, D. L. When Sugar is not so Sweet. Science 2015, 350, 275-276.
559. Glinkerman, C. M.; Boger, D. L.; Cycloadditions of 1,2,3-Triazines Bearing C5-Electron Donating Substituents: Robust Pyrimidine Synthesis. Org. Lett. 2015, 17, 4002-4005.
558. Lee, K.; Poudel, Y. B.; Glinkerman, C. M.; Boger, D. L. Total synthesis of dihydrolysergic acid and dihydrolysergol: development of a divergent synthetic strategy applicable to rapid assembly of D-ring analogs. Tetrahedron, 2015, 71 (35), 5897–5905.
557. Lee, K.; Boger, D. L. Total synthesis of (−)-kopsinine and ent-(+)-kopsinine. Tetrahedron 2015, 71 (22), 3741–3746.
556. Okano, A.; Nakayama, A.; Wu, K.; Lindsey E. A.; Schammel, A. W.; Feng, Y.; Collins K. C.; Boger, D. L. Total Syntheses and Initial Evaluation of [Ψ[C(═S)NH]Tpg4]vancomycin, Ψ[C(═NH)NH]Tpg4]vancomycin, [Ψ[CH2NH]Tpg4]vancomycin, and Their (4-Chlorobiphenyl)methyl Derivatives: Synergistic Binding pocket and Peripheral Modifications for the Glycopeptide Antibiotics. J. Am. Chem. Soc. 2015. 137, 3693–3704.
555. Sears, J. E; Boger, D. L. Total Synthesis of Vinblastine, Related Natural Products, and Key Analogues and Development of Inspired Methodology Suitable for the Systematic Study of Their Structure–Function Properties, ACC. Chem Res. 2015, 48, 653-662.
554. Anderson, E. D.; Duerfeldt, A. S.; Zhu, K.; Glinkerman, C. M.; Boger, D. L. Cycloadditions of Noncomplementary Substituted 1, 2, 3-Triazines. Org. Lett. 2014, 16, 5084-5087.
553. Duncan, K. K.; Otrubova, K.; Boger, D. L. α-Ketoheterocycle inhibitors of fatty acid amide hydrolase: Exploration of conformational constraints in the acyl side chain. Bioorg. Med. Chem. 2014, 22, 2763-2770.
552. Okano, A.; Nakayama, A.; Schammel, A. W.; Boger, D. L. Total Synthesis of [Ψ[C(═NH)NH]Tpg4] Vancomycin and its (4-Chlorobiphenyl) methyl Derivative: Impact of Peripheral Modifications on Vancomycin Analogues Redesigned for Dual D-Ala-D-Ala and D-Ala-D-Lac Binding. J. Am. Chem. Soc. 2014, 136, 13522-13525.
551. Nakayama, A.; Okano, A.; Feng, Y.; Collins, J. C.; Collins, K. C.; Walsh, C. T.; Boger, D. L. Enzymatic Glycosylation of Vancomycin Aglycon: Completion of a Total Synthesis of Vancomycin and N-and C-Terminus Substituent Effects of the Aglycon Substrate. Org. Lett. 2014, 16, 3572-3575.
550. Otrubova, K.; Srinivasan, V.; Boger, D. L. Discovery libraries targeting the major enzyme classes: The serine hydrolases. Bioorg. Med. Chem. Lett. 2014, 24, 3807-3813.
549. Sanchez-Cespedes, J.; Moyer, C. L.; Whitby, L. R.; Boger, D. L.; Nemerow, G. R. Inhibition of adenovirus replication by a trisubstituted piperazin-2-one derivative. Antiviral Res. 2014.
548. Uematsu, M.; Boger, D. L. Asymmetric Synthesis of a CBI-Based Cyclic N-Acyl O-Amino Phenol Duocarmycin Prodrug. J. Org. Chem. 2014, 79, 9699-9703.
547. C. J. Dreyton, E. D. Anderson, V. Subramanian, D. L. Boger, P. R. Thompson, Insights into the mechanism of streptonigrin-induced protein arginine deiminase inactivation, Bioorg. Med. Chem. Lett. 2014, 22, 1362–1369.
546. K. Lee, D. L. Boger, Total Syntheses of (−)-Kopsifoline D and (−)-Deoxoapodine: Divergent Total Synthesis via Late-Stage Key Strategic Bond Formation, J. Am. Chem. Soc. 2014, 136, 3312–3317.
545. K. Otrubova, B. F. Cravatt, D. L. Boger, Design, Synthesis, and Characterization of α-Ketoheterocycles That Additionally Target the Cytosolic Port Cys269 of Fatty Acid Amide Hydrolase, J. Med. Chem. 2014, 57, 1079–1089.
544. A. S. Duerfeldt, D. L. Boger, Total Syntheses of (−)-Pyrimidoblamic Acid and P-3A, J. Am. Chem. Soc. 2014, 136, 2119–2125.
543. E. L. Campbell, C. K. Skepper, K. Sankar, K. K. Duncan, D. L. Boger, Transannular Diels–Alder/1,3-Dipolar Cycloaddition Cascade of 1,3,4-Oxadiazoles: Total Synthesis of a Unique Set of Vinblastine Analogues, Org. Lett. 2013, 15, 5306–5309.
542. T. J. Barker, K. K. Duncan, K. Otrubova, D. L. Boger, Potent Vinblastine C20' Ureas Displaying Additionally Improved Activity Against a Vinblastine-Resistant Cancer Cell Line, ACS Med. Chem. Lett. 2013, 4, 985–988.
541. A. L. Wolfe, K. K. Duncan, J. P. Lajiness, K. Zhu, A. S. Duerfeldt, and D. L. Boger, A fundamental relationship between hydrophobic properties and biological activity for the duocarmycin SA class of DNA alkylating antitumor drugs: hydrophobic binding-driven-bonding, J. Med. Chem. 2013, 56, 6845–6857.
540. M. Gochin, L. R. Whitby, A. H. Phillips, and D. L. Boger, NMR-assisted computational studies of peptidometric inhibitors bound in the hydrophobic pocket of HIV-1 glycoprotein 41, J. Compt. Aids Med. Des. 2013, 27, 569–582.
539. S. Connelly, J. K. DeMartino, D. L. Boger, and I. A. Wilson, Biological and structural evaluation of 10R- and 10S-methylthio-DDACTHF reveals a new role for sulfur in inhibition of glycinamide ribonucleotide transformylase, Biochemistry 2013, 52, 5133–5144.
538. J. R. Pinchman and D. L. Boger, Investigation into the functional impact of the vancomycin C-ring aryl chloride, Bioorg. Med. Chem. Lett. 2013, 23, 4817–4819.
537. J. R. Pinchman and D. L. Boger, Probing the role of the vancomycin E-ring chloride: selective divergent synthesis and evaluation of alternatively substituted E-ring analogues, J. Med. Chem. 2013, 56, 4116–4124.
536. A. L. Wolfe, K. K. Duncan, N. K. Parelkar, D. Brown, G. A. Vielhauer, and Dale. L. Boger, Efficacious cyclic N-acyl O-amino phenol duocarmycin prodrugs, J. Med. Chem. 2013, 56, 4109–4115.
535. G. A. Vielhauer, M. Swink, N. K. Parelkar, J. P. Lajiness, A. L. Wolfe, and D. L. Boger, Evaluation of a Redcutively Activated Duocarmycin Prodrug Against Murine and Human Solid Cancers, Cancer Biol. Ther. 2013, 14, 527–536.
534. K. Otrubova, M. Brown, M. S. McCormick, G. W. Han, S. T. O'Neal, B. F. Cravatt, R. C. Stevens, A. H. Lichtman, and D. L. Boger, Rational Design of Fatty Acid Amide Hydrolase Inhibitors That Act by Covalently Bonding to Two Active Site Residues, J. Am. Chem. Soc. 2013, 135, 6289–6299.
533. T. C. Turner, K. Shibayama, and D. L. Boger, Hypervalent iodine(III)-promoted intermolecular C-C coupling of vindoline with β-ketoesters and related substrates, Org. Lett. 2013, 15, 1100–1103.
532. J. Xie, A. L. Wolfe, and D. L. Boger, Total synthesis of kopsinine, Org. Lett. 2013, 15, 868–870.
531. S. P. Breazzano, Y. B. Poudel, and D. L. Boger, A Pd(0)-mediated indole (macro)cyclization reaction, J. Am. Chem. Soc. 2013, 135, 1600–1606.
530. E. K. Leggans, K. K. Duncan, T. J. Barker, K. D. Schleicher, and D. L. Boger, A remarkable series of vinblastine analogues displaying enhanced activity and an unprecedented tubulin binding steric tolerance: C20’ urea derivatives, J. Med. Chem. 2013, 56, 628–639.
529. K. D. Schleicher, Y. Sasaki, A. Tam, D. Kato, K. K. Duncan, and D. L. Boger, Total synthesis and evaluation of vinblastine analogues containing systematic deep-seated modifications in the vindoline subunit ring system: core redesign, J. Med. Chem. 2013, 56, 483–495.
528. T. J. Barker and D. L. Boger, Fe(III)/NaBH4-mediated free radical hydrofluorination of unactivated alkenes, J. Am. Chem. Soc. 2012, 134, 13588–13591.
527. H. Gotoh, J. E. Sears, A. Eschenmoser, and D. L. Boger, New insights into the mechanism and an expanded scope of the Fe(III)-mediated vinblastine coupling reaction, J. Am. Chem. Soc. 2012, 134, 13240–13243.
526. D. L. Boger, Inhibitors of fatty acid amide hydrolase (FAAH), in Chembiomolecular Science: At the Frontier of Chemistry and Biology, Shibasaki, M., Iino, M., Osada, H., Eds., Springer, Japan; 2012, pp 37–49.
525. A. Okano, R. C. James, J. G. Pierce, J. Xie, and D. L. Boger, Silver(I)-promoted conversion of thioamides to amidines: divergent synthesis of a key series of vanomycin aglycon residue 4 amidines that clarify binding behavior to model ligands, J. Am. Chem. Soc. 2012, 134, 8790-8793.
524. A. L. Wolfe, K. K. Duncan, N. Parelkar, S. J. Weir, G. A. Vielhauer, and D. L. Boger, A novel, unusually efficacious duocarmycin carbamate prodrug that releases no residual byproduct, J. Med. Chem. 2012, 55, 5878–5886.
523. L. R. Whitby and D. L. Boger, Comprehensive peptidomimetic libraries targeting protein-protein interactions. Acc. Chem. Res. 2012, 45, 1698–1709.
522. J. P. Lajiness, W. Jiang, and D. L. Boger, Divergent total synthesis of (–)-aspidospermine and (+)-spegazzinine, Org. Lett. 2012, 14, 2078–2081.
521. L. R. Whitby, K. E. Boyle, L. Cai, M., X. Yu, M. Gochin, and D. L. Boger, Discovery of HIV fusion inhibitors targeting gp41 using a comprehensive α-helix mimetic library, Bioorg. Med. Chem. Lett.2012, 22, 2861–2865.
520. R. C. James, J. G. Pierce, A. Okano, J. Xie, and D. L. Boger, Redesign of glycopeptide antibiotics: back to the future, ACS Chem. Biol. 2012, 7, 797–804. (Invited Perspective)
519. E. K. Leggans, T. J. Barker, K. K. Duncan, and D. L. Boger, Iron(III)/NaBH4-mediated additions to unactivated alkenes: synthesis of novel C20’ vinblastine analogues, Org. Lett. 2012, 14, 1428–1431.(Ranked best in 2012 by Organic Letters)
518. K. Otrubova and D. L. Boger, α-Ketoheterocycle-based inhibitors of fatty acid amide hydrolase, ACS Chem. Neurosci. 2012, 3, 340–348. (Invited mini-review).
517. J. Xie, A. Okano, J. G. Pierce, R. C. James, S. Stamm, C. M. Crane, and D. L. Boger, Total synthesis of [Ψ[C(=S)NH]Tpg4]vancomycin aglycon, [Ψ[C(=NH)NH]Tpg4]vancomycin aglycon and related key compounds: reengineering vancomycin for dual D-Ala-D-Ala and D-Ala-D-Lac binding, J. Am. Chem. Soc. 2012, 134, 1284–1297.
516. L. Booker, S. G. Kinsey, R. A. Abdullah, J. L. Blankman, J. Z. Long, C. Ezzili, D. L. Boger, B. F. Cravatt, and A. H. Lichtman, The FAAH inhibitor PF-3845 acts in the nervous system to reverse lipopolysaccharide-induced tactile allodynia in mice, Br. J. Pharmacol. 2012, 165, 2485–2496.
515. H. Gotoh, K. K. Duncan, W. M. Robertson, and D. L. Boger, 10’-Fluorovinblastine and 10’-fluorovincristine: synthesis of a key series of modified vinca alkaloids, ACS Med. Chem. Lett. 2011, 2, 948–952.
514. S. P. Breazzano and D. L. Boger, Synthesis and stereochemical determination of complestatin A and B (neuroprotection A and B), J. Am. Chem. Soc. 2011, 133, 18495–18502.
513. J. Xie, J. G. Pierce, R. C. James, A. Okano, and D. L. Boger, A redesigned vancomycin engineered for dual D-Ala-D-Ala and D-Ala-D-Lac binding exhibits potent antimicrobial activity against vancomycin-resistant bacteria, J. Am. Chem. Soc. 2011, 133, 13946–13949.
512. E. D. Anderson and D. L. Boger, Inverse electron demand Diels-Alder reactions of 1,2,3-triazines: pronounced subsituent effects on reactivity and cycloaddition scope, J. Am. Chem. Soc. 2011, 133, 12285–12292.
511. K. Otrubova, C. Ezzili, and D. L. Boger, The discovery and development of inhibitors of fatty acid amide hydrolase (FAAH), Bioorg. Med. Chem. Lett. 2011, 21, 4674–4685. (Invited BMCL Digest)
510. L. R. Whitby, Y. Ando, V. Setola, P. K. Vogt, B. L. Roth, and D. L. Boger, Design, synthesis and validation of a β-turn mimetic library targeting protein–protein and peptide–receptor interactions, J. Am. Chem. Soc. 2011, 133, 10184–10194.
509. E. D. Anderson and D. L. Boger, Scope of the inverse electron demand Diels–Alder reactions of 1,2,3-triazine, Org. Lett. 2011, 13, 2492–2494.
508. C. Ezzili, M. Mileni, N. McGlinckey, J. Z. Long, S. G. Kinsey, D. G. Hochstatter, R. C. Stevens, A. H. Lichtman, B. F. Cravatt, E. J. Bilsky, and D. L. Boger, Reversible competitive α-ketoheterocycle inhibitors of fatty acid amide hydrolase containing additional conformational constraints in the acyl side chain: orally active, long acting analgesics, J. Med. Chem. 2011, 54, 2805–2822.
507. M. Mileni, J. Garfunkle, C. Ezzili, B. F. Cravatt, R. C. Stevens, and D. L. Boger, Fluoride-mediated capture of a noncovalent bound state of a reversible covalent enzyme inhibitor: X-ray crystallographic analysis of an exceptionally potent α-ketoheterocycle inhibitor of fatty acid amide hydrolase, J. Am. Chem. Soc. 2011, 133, 4092–4100.
506. C. J. Thomas, H. E. Casquilho, J. York, D. L. DeCamp, D. Dai, E. B. Petrilli, D. L. Boger, R. A. Slayden, S. M. Amberg, S. R. Sprang, and J. H. Nunberg, A specific interaction of small molecule entry inhibitors with the envelope glycoprotein complex of the Junin hemorrhagic fever arenavirus, J. Biol. Chem. 2011, 286, 6192–6200.
505. J. P. Lajiness and D. L. Boger, Asymmetric synthesis of 1,2,9,9a-tetrahydrocyclopropa[c]benzo[e]indol-4-one (CBI), J. Org. Chem. 2011, 76, 583–587.
504. J. P. Lajiness, W. M. Robertson, I. Dunwiddie, M. A. Broward, G. A. Vielhauer, S. J. Weir, and D. L. Boger, Design, synthesis, and evaluation of CC-1065 and duocarmycin O-aminophenol prodrugs subject to tunable reductive activation, J. Med. Chem. 2010, 53, 7731–7738.
503. A. Tam, H. Gotoh, W. M. Robertson, and D. L. Boger, Catharanthine C16 substituent effects on the biomimetic coupling with vindoline: preparation and evaluation of a key series of vinblastine analogues, Bioorg. Med. Chem. Lett. 2010, 20, 6408–6410.
502. J. P. Lajiness and D. L. Boger, Synthesis and characterization of a cyclobutane duocarmycin derivative incorporating the CbBI (1,2,10,11-tetrahydro-9H-cyclobuta[c]benzo[e]indol-4-one) alkylation subunit, J. Am. Chem. Soc. 2010, 132, 13936-13940.
501. C. M. Crane, J. G. Pierce, S. S. F. Leung, J. Tirado-Rives, W. L. Jorgensen, and D. L. Boger, Synthesis and evaluation of selected key methyl ether derivatives of vancomycin aglycon, J. Med. Chem.2010, 53, 7229-7235.
500. Y. Sasaki, D. Kato, and D. L. Boger, Asymmetric total synthesis of vindorosine, vindoline, and key vinblastine analogues, J. Am. Chem. Soc. 2010, 132, 13533-13544.
499. G. Ambrus, L. R. Whitby, E. L. Singer, O. Trott, E. Choi, A. J. Olson, D. L. Boger, and L. Gerace, Small molecule peptidomimetic inhibitors of importin α/β mediated nuclear transport, Bioorg. Med. Chem. 2010, 10, 7611-7620.
498. C. Ezzili, K. Otrubova, and D. L. Boger, Fatty acid amide signaling molecules, Bioorg. Med. Chem. Lett. 2010, 20, 5959-5968. (Invited BMCL Digest).
497. P. R. Patel and D. L. Boger, Intramolecular Diels-Alder reaction of cyclopropenone ketals, Org. Lett. 2010, 12, 3540-3543.
496. C. P. Burke, M. R. Swingle, R. E. Honkanen, and D. L. Boger, Total synthesis and evaluation of phostriecin and key structural analogues, J. Org. Chem. 2010, 75, 7505-7513. (Feature Article).
495. P. R. Patel and D. L. Boger, Intramolecular [1+2] and [3+2] cycloaddition reactions of cyclopropenone ketals, J. Am. Chem. Soc. 2010, 132, 8527-8529.
494. P. Va, E. L. Campbell, W. M. Robertson, and D. L. Boger, Total synthesis and evaluation of a key series of C5-substituted vinblastine derivatives, J. Am. Chem. Soc. 2010, 132, 8489-8495.
493. H. Shimamura, S. P. Breazzano, J. Garfunkle, F. S. Kimball, J. D. Trzupek, and D. L. Boger, Total synthesis of complestatin: development of a Pd(0)-mediated indole annulation for macrocyclization, J. Am. Chem. Soc. 2010, 132, 7776-7783.
492. W. M. Robertson, D. B. Kastrinsky, I. Hwang, and D. L. Boger, Synthesis and evaluation of a series of C5'-substituted duocarmycin SA analogs, Bioorg. Med. Chem. Lett. 2010, 20, 2722--2725.
491. J. S. Oakdale and D. L. Boger, Total synthesis of lycogarubin C and lycogalic acid, Org. Lett. 2010, 12, 1132-1134.
490. D. Kato, Y. Sasaki, and D. L. Boger, Asymmetric total synthesis of vindoline, J. Am. Chem. Soc. 2010, 132, 3685-3687.
489. K. E. Boyle, K. S. MacMillan, D. A. Ellis, J. P. Lajiness, W. M. Robertson, and D. L. Boger, Synthesis and evaluation of duocarmycin SA analogs incorporating the methyl 1,2,8,8a-tetrahydrocyclopropa[c]oxazolo[2,3-e]indole-4-one-6-carboxylate (COI) alkylation subunit,, Bioorg. Med. Chem. Lett. 2010, 20, 1854-1857.
488. E. L. Campbell, A. M. Zuhl, C. M. Liu, and D. L. Boger, Total synthesis of (+)-fendleridine (aspidoalbidine) and (+)-1-acetylaspidoalbidine, J. Am. Chem. Soc. 2010, 132, 3009-3012.
487. C. P. Burke, N. Haq, and D. L. Boger, Total synthesis, assignment of relative and absolute stereochemistry, and structural reassignment of phostriecin (aka sultriecin), J. Am. Chem. Soc. 2010, 132, 2157-2159.
486. M. J. Giffin, R. Muller, J. Savage, Y. C. Lin, S. Hong, W. Jin, L. R. Whitby, J. H. Elder, D. L. Boger, and R. E. Torbett, Identification of broad-based HIV-1 protease inhibitors from combinatorial libraries, ChemBioChem. 2010, 429, 527-532.
485. J. Garfunkle, F. S. Kimball, J. D. Trzupek, S. Takazawa, H. Shimamura, M. Tomishima, and D. L. Boger, Total synthesis of chloropeptin II (complestatin) and chloropeptin I, J. Am. Chem. Soc. 2009,131, 16036-16038.
484. K. S. MacMillan, J. P. Lajiness, C. Lopez-Cara, R. Romagnoli, W. M. Robertson, I. Hwang, P. G. Baraldi, and D. L. Boger, Synthesis and evaluation of a thio analogue of duocarmycin SA, Bioorg. Med. Chem. Lett. 2009, 19, 6962-6965.
483. M. Mileni, J. Garfunkle, C. Ezzili, F. S. Kimball, B. F. Cravatt, R. C. Stevens, and D. L. Boger, X-ray crystallographic analysis of ?-ketoheterocycle-based inhibitors bound to a humanized variant of fatty acid amide hydrolase, J. Med. Chem. 2010, 52, 230-240.
482. J. Shi, J. S. Stover, L. R. Whitby, P. K. Vogt, and D. L. Boger, Small molecule inhibitors of Myc/Max dimerization and Myc-induced cell transformation, Bioorg. Med. Chem. Lett. 2009, 19, 6038-6041.
481. X. Fang, J. Nam, D. Shin, Y. Rew, D. L. Boger, and S. Walker, Functional and biochemical analysis of a key series of ramoplanin analogues, Bioorg. Med. Chem. Lett. 2009, 19, 6189-6191.
480. M. B. Swingle, L. Amable, B. G. Lawhorn, S. B. Buck, C. P. Burke, P. Ratti, K. L. Fisher, D. L. Boger, and R. E. Honkanen, Structure-activity relationship studies of fostriecin, cytostatin, and key analogs with PP1, PP2A, PP5 and (β12 - β13)-chimeras (PP1/PP2A and PP5/PP2A) provide further insight into inhibitory actions of fostriecin family inhibitors, J. Pharmacol. Exp. Ther. 2009, 331, 45-53.
479. K. S. MacMillan and D. L. Boger, Fundamental relationships between structure, reactivity, and biological activity for the duocarmycins and CC-1065, J. Med Chem. 2009, 52, 5771-5780.
478. S. G. Kinsey, J. Z. Long, S. T. O'Neal, R. A. Abdulla, J. L. Poklis, D. L. Boger, B. F. Cravatt, and A. H. Lichtman, Blockade of endocannabinoid-degrading enzymes attenuates neuropathic pain, J. Pharmacol. Exp. Ther. 2009, 330, 902-910.
477. V. Subramanian, R. M. Williams, D. L. Boger, and K. Luger, Methods to characterize the effect of DNA-modifying compounds on nucleosomal DNA, in Methods in Molecular Biology, Drug-DNA Interactions 2nd Ed., Humana Press, Totowa, NJ, 2009, 173-192.
476. M. Mileni, J. Garfunkle, J. K. DeMartino, B. F. Cravatt, D. L. Boger, and R. C. Stevens, Binding and inactivation mechanism of a humanized fatty acid amide hydrolase by -ketoheterocycle inhibitors revealed from co-crystal structures, J. Am. Chem. Soc. 2009, 131, 10497-10506.
475. L. R. Whitby, A. M. Lee, S. Kunz, M. B. A. Oldstone, and D. L. Boger, Characterization of Lassa virus cell entry inhibitors: determination of the active enantiomer by asymmetric synthesis, Bioorg. Med. Chem. Lett. 2009, 19, 3771-3774. Symposium-in-Print honoring C. Barbas, 2009 Tetrahedron Young Investigator Award.
474. K. E. Hauschild, J. S. Stover, D. L. Boger, and A. Z. Ansari, CSI-FID: high throughput label-free detection of DNA binding molecules, Bioorg. Med. Chem. Lett. 2009, 19, 3779-3782. Symposium-in-Print honoring C. Barbas, 2009 Tetrahedron Young Investigator Award.
473. D. L. Boger and H. Ishikawa, Total synthesis of vindoline and related alkaloids, J. Syn. Org. Chem., Jpn. 2009, 67, 123-133.
472. A. Shaginian, L. R. Whitby, S. Hong, I. Hwang, B. Faroogi, J. Chen, M. Searcey, P. K. Vogt, and D. L. Boger, Design, synthesis, and evaluation of an α-helix mimetic library targeting protein-protein interactions, J. Am. Chem. Soc. 2009, 131, 5564-5572.
471. C. M. Gauss, A. Hamasaki, J. P. Parrish, K. S. MacMillan, T. J. Rayl, I. Hwang, and D. L. Boger, Synthesis and preliminary evaluation of duocarmycin analogues incorporating the 1,2,11,11a-tetrahydrocyclopropa [c]naphtho[2,3-e]indol-4-one (CNI) and 1,2,11,11a-tetrahydrocyclopropa[c]naphtho[1,2-e]indol-4-one (iso-CNI) alkylation subunits, Tetrahedron 2009, 65, 6591-6599. Symposium-in-Print honoring L. Overman, 2009 Tetrahedron Award.
470. J. E. Schlosburg, D. L. Boger, B. F. Cravatt, and A. H. Lichtman, Endocannabinoid modulation of scratching response in an acute allergenic model: new prospective neural therapeutic target for pruritus, J. Pharmacol. Exp. Ther. 2009, 329, 314-323.
469. H. Ishikawa, D. A. Colby, S. Seto, P. Va, A. Tam, H. Kakei, T. J. Rayl, I. Hwang, and D. L. Boger, Total synthesis of vinblastine, vincristine, related natural products and key structural analogues, J. Am. Chem. Soc. 2009, 131, 4904-4916.
468. R. C. Clark, S. Y. Lee, I. Hwang, M. Searcey, and D. L. Boger, Evaluation of chlorofusin, its seven chromophore diastereomers, and key analogues, Tetrahedron Lett. 2009, 50, 3151-3153. Symposium-in-Print commemorating the 50th Anniversary of Tetrahedron Letters.
467. C. M. Crane and D. L. Boger, Synthesis and evaluation of vancomycin aglycon analogues that bear modifications in the N-terminus D-leucyl amino acid, J. Med. Chem. 2009, 52, 1471-1476.
466. J. S. Stover, J. Shi, W. Jin, P. K. Vogt, and D. L. Boger, Discovery of inhibitors of aberrant gene transcription from libraries of DNA binding molecules: inhibition of LEF-1 mediated gene transcription and oncogenic transformation, J. Am. Chem. Soc. 2009, 131, 3342-3348.
465. R. C. Clark, S. Y. Lee, M. Searcey, and D. L. Boger, The isolation, total synthesis and structure elucidation of chlorofusin, a natural product inhibitor of the p53-MDM2 protein-protein interaction,Natural Prod. Rep. 2009, 26, 465-477.
464. K. S. MacMillan, T. Nguyen, I. Hwang, and D. L. Boger, Total synthesis and evaluation of iso-duocarmycin SA and iso-yatakemycin, J. Am. Chem. Soc. 2009, 131, 1187-1194.
463. S. Y. Lee and D. L. Boger, Synthesis of the chlorofusin cyclic peptide, Tetrahedron 2009, 65, 3281-3284. Symposium-in-Print in honor of Justin Du Bois, 2008 Tetrahedron Young Investigator Award.
462. J. K. DeMartino and D. L. Boger, Glycinamide ribonucleotide transformylase (GAR Tfase) as a target for cancer therapy, Drugs Fut. 2008, 33, 969–979.
461. K. S. MacMillan and D. L. Boger, An alternative spirocyclization for duocarmycin SA, J. Am. Chem. Soc. 2008, 130, 16521–16523.
460. J. K. DeMartino, I. Hwang, S. Connolly, I. A. Wilson, and D. L. Boger, Asymmetric synthesis of inhibitors of glycinamide ribonucleotide transformylase, J. Med. Chem. 2008, 51, 5441–5448.
459. J. K. DeMartino, J. Garfunkle, D. G. Hochstatter, B. F. Cravatt, and D. L. Boger, Exploration of a fundamental substituent effect of a-ketoheterocycle enzyme inhibitors: potent and selective inhibitors of fatty acid amide hydrolase, Bioorg. Med. Chem. Lett. 2008, 18, 5842–5846. Symposium-in-print honoring B. F. Cravatt, 2008 Tetrahedron Young Investigator Award.
458. I. Slaymaker, M. Bracey, M. Mileni, J. Garfunkle, B. F. Cravatt, D. L. Boger, and R. C. Stevens, Correlation of inhibitor effects on enzyme activity and thermal stability for the integral membrane protein fatty acid amide hydrolase, Bioorg. Med. Chem. Lett. 2008, 18, 5847–5850. Symposium-in-print honoring B. F. Cravatt, 2008 Tetrahedron Young Investigator Award.
457. A. M. Lee, J. M. Rojek, C. F. Spiropoulou, A. Gunderson, W. Jin, A. Shaginian, J. York, J. H. Nunberg, D. L. Boger, M. B. A. Oldstone, and S. Kunz, Unique small molecule entry inhibitors of hemorrahagic fever arenaviruses, J. Biol. Chem. 2008, 283, 18734–18742.
456.R. C. Clark, S. Y. Lee, and D. L. Boger, Total synthesis of chlorofusin, its seven chromophore diastereomers, and key partial structures, J. Am. Chem. Soc. 2008, 130, 12355–12369.
455. J. Garfunkle, C. Ezzili, T. J. Rayl, D. G. Hochstatter, I. Hwang, and D. L. Boger, Optimization of the central heterocycle of a-ketoheterocycle inhibitors of fatty acid amide hydrolase, J. Med. Chem.2008, 51, 4392–4403.
454. X. Lu, P. K. Vogt, D. L. Boger, and J. Lunec, Disruption of the MYC transcriptional function by a small molecule antagonist of Myc/Max dimerization, Oncol. Rep. 2008, 19, 825–830.
453. S. Di Micco, D. L. Boger, R. Riccio, and G. Bifulco, Structural features of the (+)-yatakemycin–d(GACTAATTGAC)-(GTCAATTAGTC) complex. Quantum mechanical calculation of NMR parameters as a tool for the characterization of ligand-DNA interactions, Eur. J. Org. Chem. 2008, 2454–2462.
452. H. Ishikawa, D. A. Colby, and D. L. Boger, Direct Coupling of catharanthine and vindoline to provide vinblastine: total synthesis of (+)- and ent-(-)-vinblastine, J. Am. Chem. Soc. 2008, 130, 420–421.
451. F. S. Kimball, F. A. Romero, C. Ezzili, J. Garfunkle, T. J. Rayl, D. G. Hochstatter, I. Hwang, and D. L. Boger, Optimization of ?-ketooxazole inhibitors of fatty acid amide hydrolase, J. Med. Chem. 2008, 51, 937–947.
450. M. S. Tichenor and D. L. Boger, Yatakemycin: total synthesis, DNA alkylation, and biological properties, Natural Prod. Rep. 2008, 25, 220–226.
449. W. Jin, J. D. Trzupek, T. J. Rayl, M. A. Broward, G. A. Vielhauer, S. J. Weir, I. Hwang, and D. L. Boger, A unique class of duocarmycin and CC-1065 analogues subject to reductive activation, J. Am. Chem. Soc. 2007, 129, 15391–15397.
448. M. S. Tichenor, K. S. MacMillan, J. S. Stover, S. E. Wolkenberg, M. G. Pavani, L. Zanella, A. N. Zaid, G. Spalluto, T. J. Rayl, I. Hwang, P. G. Baraldi, and D. L. Boger, Rational design, synthesis, and evaluation of key analogues of CC-1065 and the duocarmycins, J. Am. Chem. Soc. 2007, 129, 14092–14099.
447. M. S. Tichenor, K. S. MacMillan, J. D. Trzupek, T. J. Rayl, I. Hwang, and D. L. Boger, Systematic exploration of the structural features of yatakemycin impacting DNA alkylation and biological activity,J. Am. Chem. Soc. 2007, 129, 10858–10869.
446. S. Y. Lee, R. C. Clark, and D. L. Boger, Total synthesis, stereochemical reassignment, and absolute configuration of chlorofusin, J. Am. Chem. Soc. 2007, 129, 9860–9861.
445. L. Xu, Y. Chong, I. Hwang, A. D’Onofrio, K. Amore, G. P. Beardsley, C. Li, A. J. Olson, D. L. Boger, and I. A. Wilson, Structure-based design, synthesis, evaluation, and crystal structures of transition state analogue inhibitors of inosine monophosphate cyclohydrolase, J. Biol. Chem. 2007, 282, 13033–13046.
444. J. Nam, D. Shin, Y. Rew, and D. L. Boger, Alanine scan of [L-Dap2]ramoplanin A2 aglycon: assessment of the importance of each residue, J. Am. Chem. Soc. 2007, 129, 8747–8755.
443. C. Hardouin, M. J. Kelso, F. A. Romero, T. J. Rayl, D. Leung, I. Hwang, B. F. Cravatt, and D. L. Boger, Structure–activity relationships of the a-ketooxazole inhibitors of fatty acid amide hydrolase, J. Med. Chem. 2007, 50, 3359–3368.
442. L. M. Eubanks, M. S. Hixon, W. Jin, S. Hong, C. M. Clancy, W. H. Tepp, C. J. Malizio, M. C. Goodnough, J. T. Barbieri, E. A. Johnson, D. L. Boger, T. J. Dickerson, and K. D. Janda, High throughput identification of botulinum neurotoxin A antagonists: an in vitro and in vivo disconnect, Proc. Natl. Acad. Sci. U.S.A. 2007, 104, 2602–2607
441. F. A. Romero, W. Du, I. Hwang, T. J. Rayl, F. S. Kimball, D. Leung, H. S. Hoover, R. L. Apodaca, J. G. Breitenbucher, B. F. Cravatt, and D. L. Boger, Potent and selective a-ketoheterocycle-based inhibitors of the anandamide and oleamide catabolizing enzyme, fatty acid amide hydrolase, J. Med. Chem. 2007, 50, 1058–1068.
440. H. Ishikawa and D. L. Boger, Total synthesis of (–)- and ent-(+)-4-desacetoxy-5-desethylvindoline, Heterocycles 2007, 72, 95–102. Commemorative issue honoring Y. Kishi on his 70th birthday.
439. B. G. Lawhorn, S. B. Boga, S. E. Wolkenberg, D. A. Colby, C.-M. Gauss, M. R. Swingle, L. Amable, R. E. Honkanen, and D. L. Boger, Total synthesis of cytostatin, its C10–C11 diastereomers and additional key analogues: impact on PP2A inhibition, J. Am. Chem. Soc. 2006, 129, 16720–16732.
438. M. S. Tichenor, J. D. Trzupek, D. B. Kastrinsky, F. Shiga, I. Hwang, and D. L. Boger, Asymmetric total synthesis of (+)- and ent-(–)-yatakemycin and duocarmycin SA: evaluation of yatakemycin key partial structures and its unnatural enantiomer, J. Am. Chem. Soc. 2006, 128, 15683–15696.
437. F. A. Romero, I. Hwang, and D. L. Boger, Delineation of a fundamental a-ketoheterocycle substituent effect for use in the design of enzyme inhibitors, J. Am. Chem. Soc. 2006, 128, 14004–14005.
436. B. G. Lawhorn, S. B. Boga, S. E. Wolkenberg, and D. L. Boger, Total synthesis of cytostatin, Heterocycles 2006, 70, 65–70. Commemorative issue honoring S. Weinreb on his 65th birthday.
435. M. J. Schnermann, F. A. Romero, I. Hwang, E. Nakamaru-Ogiso, T. Yagi, and D. L. Boger, Total synthesis of piericidin A1 and B1 and key analogues, J. Am. Chem. Soc. 2006, 128, 11799–11807.
434. H. Ishikawa, G. I. Elliott, J. Velcicky, Y. Choi, and D. L. Boger, Total synthesis of (–)- and ent-(+)-vindoline and related alkaloids, J. Am. Chem. Soc. 2006, 128, 10596–10612.
433. G. I. Elliott, J. R. Fuchs, B. S. J. Blagg, H. Ishikawa, H. Tao, Z.-Q. Yuan, and D. L. Boger, Intramolecular Diels–Alder/1,3-dipolar cycloaddition cascade of 1,3,4-oxadiazoles, J. Am. Chem. Soc. 2006,128, 10589–10595.
432. J. K. DeMartino, I. Hwang, L. Xu, I. A. Wilson, and D. L. Boger, Discovery of a potent, non-polyglutamatable inhibitor of glycinamide ribonucleotide transformylase, J. Med. Chem. 2006, 49, 2998–3002.
431. B. M. Crowley and D. L. Boger, Total synthesis and evaluation of [Ψ(CH2NH)Tpg4]vancomycin aglycon: reengineering vancomycin for dual D-Ala-D-Ala and D-Ala-D-Lac binding, J. Am. Chem. Soc.2006, 128, 2885–2892.
430. R. C. Clark, S. S. Pfeiffer, and D. L. Boger, Diastereoselective Diels–Alder reactions of N-sulfonyl-1-aza-1,3-butadienes with optically active enol ethers: an asymmetric variant of the 1-azabutadiene Diels–Alder reaction, J. Am. Chem. Soc. 2006, 128, 2587–2593.
429. X. Fang, K. Tiyanont, Y. Zhang, J. Wanner, D. L. Boger, and S. Walker, The mechanism of action of ramoplanin and enduracidin, Mol. BioSystems 2006, 2, 69–76.
428. J. D. Trzupek, J. M. Gottesfeld, and D. L. Boger, Sequence-selective alkylation of duplex DNA in nucleosome core particles with duocarmycin SA and yatakemycin, Nature Chem. Biol. 2006, 2, 79–82.
427. A. Hamasaki, R. Ducray, and D. L. Boger, Two novel 1,2,4,5-tetrazines that participate in inverse electron demand Diels–Alder reactions with an unexpected regioselectivity, J. Org. Chem. 2006, 71, 185–193.
426. G. I. Elliott, J. Velcicky, H. Ishikawa, Y. Li, and D. L. Boger, Total synthesis of (–)- and ent-(+)-vindorosine: tandem intramolecular Diels–Alder/1,3-dipolar cycloaddition of 1,3,4-oxadiazoles, Angew. Chem. Int. Ed. 2006, 45, 620–622.
425. C. R. W.Guimarães, D. L. Boger, and W. L Jorgensen, Elucidation of fatty acid amide hydrolase inhibition by potent a-ketoheterocycle derivatives from Monte Carlo simulations, J. Am. Chem. Soc. 2005, 127, 17377–17384.
424. M. J. Schnermann and D. L. Boger, Total synthesis of piercidin A1 and B1 J, Am. Chem. Soc. 2005, 127, 15704–15705.
423. Y. Choi, H. Ishikawa, J. Velcicky, G. I. Elliott, M. M. Miller, and D. L. Boger, Total synthesis of (–)- and ent-(+)-vindoline, Org. Lett. 2005, 7, 4539–4542.
422. A. Hamasaki, J. M. Zimpleman, I. Hwang, and D. L. Boger, Total synthesis of ningalin D, J. Am. Chem. Soc. 2005, 127, 10767–10770.
421. K. J. Capps, J. Humiston, R. Dominique, I. Hwang, and D. L. Boger, Discovery of a new class of AICAR Tfase inhibitors that disrupt requisite enzyme dimerization, Bioorg. Med. Chem. Lett. 2005, 15, 2840–2844.
420. Z. Yuan, H. Ishikawa, and D. L. Boger, Total synthesis of natural (+)- and ent-(–)-4-desacetoxy-6,7-dihydrovindorosine and natural and ent-minovine: oxadiazole tandem intramolecular Diels–Alder/1,3-dipolar cycloaddition reaction, Org. Lett. 2005, 7, 741–745. Correction: Org. Lett. 2005, 7, 2079.
419. T. -C. Chou, Y. Guan, D. R. Soenen, S. J. Danishefsky, and D. L. Boger, Potent reversal of multidrug resistance by ningalin and its use in drug combinations against human colon carcinoma xenografts in nude mice Cancer Chemother, Pharmacol. 2005, 56, 379–390.
418. D. Leung, W. Du, C. Hardouin, H. Cheng, I. Hwang, B. F. Cravatt, and D. L. Boger, Discovery of a class of exceptionally potent and selective inhibitors of fatty acid amide hydrolase enlisting proteome-wide selectivity screening: concurrent optimization of an enzyme inhibitor potency and selectivity, Bioorg. Med. Chem. Lett. 2005, 15, 1423–1428.
417. H. Cheng, I. Hwang, Y. Chong, A. Tavassoli, M. E. Webb, Y. Zhang, I. A. Wilson, S. J. Benkovic, and D. L. Boger, Synthesis and biological evaluation of N-{4-[5-(2,4-diamino-6-oxo-1,6-dihydropyrimidin-5-yl)-2-(2,2,2-trifluoroacetyl)pentyl]benzoyl}-L-glutamate as a potential inhibitor of GAR Tfase and the de novo purine biosynthetic pathway, Bioorg. Med. Chem. 2005, 13, 3593–3599.
416. Y. Chong, I. Hwang, A. Tavassoli, Y. Zhang, I. A. Wilson, S. J. Benkovic, and D. L. Boger, Synthesis and biological evaluation of a- and g-carboxamide derivatives of 10-CF3CO-DDACTHF, Bioorg. Med. Chem. 2005, 13, 3587–3592.
415. H. Cheng, Y. Chong, I. Hwang, A. Tavassoli, Y. Zhang, I. A. Wilson, S. J. Benkovic, and D. L. Boger, Design, synthesis, and evaluation of 10-methanesulfonyl-DDACTHF, 10-methanesulfonyl-5-DACTHF, and 10-methylthio-DDACTHF as potent inhibitors of GAR Tfase and the de novo purine biosynthetic pathway, Bioorg. Med. Chem. 2005, 13, 3577–3585.
414. W. C. Tse and D. L. Boger, A fluorescent intercalator displacement (FID) assay for establishing DNA binding selectivity and affinity, Curr. Protocols Nucleic Acid Chem. 2005, in press.
413. S. Walker, J. Helm, Y. Hu, L. Chen, Y. Rew, D. Shin, and D. L. Boger, Chemistry and biology of ramoplanin: a lipoglycodepsipeptide with potent antibiotic activity, Chem. Rev. 2005, 105, 449–476.
412. D. L. Boger, H. Miyauchi, W. Du, C. Hardouin, R. A. Fecik, H. Cheng, I. Hwang, M. P. Hedrick, D. Leung, O. Acevedo, C. R. W. Guimar�es, W. L. Jorgensen, and B. F. Cravatt, Discovery of a potent, selective, and efficacious class of reversible a-ketoheterocycle inhibitors of fatty acid amide hydrolase effective as analgesics, J. Med. Chem. 2005, 48, 1849–1856. (Paul Janssen Memorial Issue)
411. W. Du, C. Hardouin, H. Cheng, I. Hwang, and D. L. Boger, Heterocyclic sulfoxide and sulfone inhibitors of fatty acid amide hydrolase, Bioorg. Med. Chem. Lett. 2005, 15, 103–106.
410. H. Tao, I. Hwang, and D. L. Boger, Multidrug resistance reversal activity of permethyl ningalin B amide derivatives, Bioorg. Med. Chem. Lett. 2004, 14, 5979–5981.
409. J. P. Parrish, J. D. Trzupek, T. V. Hughes, I. Hwang, and D. L. Boger, Synthesis and evaluation of N-aryl and N-alkenyl CBI derivatives, Bioorg. Med. Chem. 2004, 12, 5845–5856.
408. A. H. Lichtman, D. Leung, C. C. Shelton, A. Saghatelian, C. Hardouin, D. L. Boger, and B. F. Cravatt, Reversible inhibitors of fatty acid amide hydrolase that promote analgesia: evidence for an unprecedented combination of potency and selectivity, J. Pharmacol. Exp. Ther. 2004, 311, 441–448.
407. Y. -W. Ham and D. L. Boger, A powerful selection assay for mixture libraries of DNA alkylating agents, J. Am. Chem. Soc. 2004, 126, 9194–9195.
406. M. S. Tichenor, D. B. Kastrinsky, and D. L. Boger, Total synthesis, structure revision, and absolute configuration of (+)-yatakemycin, J. Am. Chem. Soc. 2004, 126, 8396–8398.
405. P. S. Lee, W. Du, D. L. Boger, and W. L. Jorgensen, Energetic preferences for α,β versus β,γ unsaturation, J. Org. Chem. 2004, 69, 5448–5453.
404. L. Chen, Y. Yuan, J. S. Helm, Y. Hu, Y. Rew, D. Shin, D. L. Boger, and S. Walker, Dissecting ramoplanin: mechanistic analysis of synthetic ramoplanin analogues as a guide to the design of improved antibiotics, J. Am. Chem. Soc. 2004, 126, 7462–7463.
403. D. Shin, Y. Rew, and D. L. Boger, Total synthesis and structure of the ramoplanin A1 and A3 aglycons: two minor components of the ramoplanin complex, Proc. Natl. Acad. Sci. U.S.A. 2004, 101, 11977–11979.
402. A. M. Lillo, C. Sun, C. Gao, H. Ditzel, J. Parrish, C.-M. Gauss, J. Moss, B. Felding-Habermann, P. Wirsching, D. L. Boger and K. D. Janda, A human single-chain antibody specific for integrin a3b1capable of cell internalization and delivery of antitumor agents, Chem. Biol. 2004, 11, 897–906.
401. B. M. Crowley, Y. Mori, C. C. McComas, D. Tang, and D. L. Boger, Total synthesis of the ristocetin aglycon, J. Am. Chem. Soc. 2004, 126, 4310–4317.
400. D. B. Kastrinsky and D. L. Boger, Effective asymmetric synthesis of CBI, J. Org. Chem. 2004, 69, 2284–2289.
399. Y. Rew, D. Shin, I. Hwang, and D. L. Boger, Total synthesis and examination of three key analogues of ramoplanin: a lipoglycodepsipeptide with potent antibiotic activity, J. Am. Chem. Soc. 2004, 126, 1041–1043.
398. J. P. Parrish, T. V. Hughes, I. Hwang, and D. L. Boger, Establishing the parabolic relationship between reactivity and activity for derivatives and analogues of the duocarmycin and CC-1065 alkylation subunits, J. Am. Chem. Soc. 2004, 126, 80–81.
397. W. C. Tse and D. L. Boger, A fluorescent intercalator displacement (FID) assay for establishing DNA binding selectivity and affinity, Acc. Chem. Res. 2004, 37, 61–69.
396. W. C. Tse and D. L. Boger, Sequence-selective DNA recognition: natural products and nature’s lessons, Chem. Biol. 2004, 11, 1607–1617.
395. P. Desai, S. S. Pfeiffer, and D. L. Boger, Synthesis of the chlorofusin cyclic peptide: assignment of Asn stereochemistry, Org. Lett. 2003, 5, 5047–5050.
394. S. B. Buck, C. Hardouin, S. Ichikawa, D. R. Soenen, C.-M. Gauss, I. Hwang, M. R. Swingle, K. M. Bonness, R. E. Honkanen, and D. L. Boger, Fundamental role of the fostriecin unsaturated lactone and implications for selective protein phosphatase inhibition, J. Am. Chem. Soc. 2003, 125, 15694–15695.
393. C. Bassarello, P. Cimino, G. Bifulco, D. L. Boger, J. A. Smith, W. J. Chazin, and L. G. Paloma, NMR structure of the (+)-CPI-indole/d(GACTAATTGAC)-d(GTCAATTAGTC) covalent complex,ChemBioChem 2003, 4, 1188–1193.
392. J. P. Parrish, D. B. Kastrinsky, Inkyu Hwang, and D. L. Boger, Synthesis and evaluation of duocarmycin and CC-1065 analogues incorporating the 1,2,9,9a-tetrahydro-cyclopropa[c]benz[e]-3-azaindole-4-one (CBA) alkylation subunit, J. Org. Chem. 2003, 68, 8984–8990.
391. W. C. Tse, T. Ishii, and D. L. Boger, Comprehensive high resolution analysis of hairpin polyamides utilizing a fluorescent intercalator displacement (FID) assay, Bioorg. Med. Chem. 2003, 11, 4479–4486.
390. Y.-W. Ham, W. C. Tse, and D. L. Boger, High resolution assessment of protein DNA binding affinity and selectivity utilizing a fluorescent intercalator displacement (FID) assay, Bioorg. Med. Chem. Lett. 2003, 13, 3805–3807.
389. B. K. S. Yeung, W. C. Tse, and D. L. Boger, Determination of binding affinities of triplex forming oligonucleotides using a fluorescent intercalator displacement (FID) assay, Bioorg. Med. Chem. Lett.2003, 13, 3801–3804.
388. J. P. Parrish, D. B. Kastrinsky, S. E. Wolkenberg, Y. Igarashi, and D. L. Boger, DNA alkylation properties of yatakemycin, J. Am. Chem. Soc. 2003, 125, 10971–10976.
387. C. C. McComas, B. M. Crowley, and D. L. Boger, Partitioning the loss in vancomycin binding affinity for D-Ala-D-Lac into lost H-bond and repulsive lone pair contributions, J. Am. Chem. Soc. 2003,125, 9314–9315.
386. J. P. Parrish, D. B. Kastrinsky, and D. L. Boger, Synthesis and x-ray analysis of an unprecedented and stable 2-aza-4,4-spirocyclopropacyclohexadienone, Org. Lett. 2003, 5, 2577–2579.
385. B. K. S. Yeung and D. L. Boger, Synthesis of isochrysohermidin–distamycin hybrids, J. Org. Chem. 2003, 68, 5249–5253.
384. C. C. McComas, B. M. Crowley, I. Hwang, and D. L. Boger, Synthesis and evaluation of methyl ether derivatives of the vancomycin, teicoplanin, and ristocetin aglycon methyl ethers, Bioorg. Med. Chem. Lett. 2003, 13, 2933–2936. Symposium-in-print: Recent Advances in Antibiotics.
383. D. L. Boger, J. Desharnais, and K. Capps, Solution-phase combinatorial libraries: modulating cellular signaling by targeting protein–protein or protein–DNA interactions, Angew. Chem. Int. Ed. 2003,42, 4138–4176.
382. J. Desharnais, I. Hwang, Y. Zhang, A. Tavassoli, J. Baboval, S. J. Benkovic, I. A. Wilson, and D. L. Boger, Design, synthesis, and biological evaluation of 10-CF3CO-DDACTHF analogues and derivatives as inhibitors of GAR Tfase and the de novo purine biosynthetic pathway, Bioorg. Med. Chem. 2003, 11, 4511–4521.
381. T. H. Marsilje, M. P. Hedrick, J. Desharnais, K. Capps, A. Tavassoli, Y. Zhang, I. A. Wilson, S. J. Benkovic, and D. L. Boger, 10-(2-Benzoxazolcarbonyl)-5,10-dideaza-acyclic-5,6,7,8-tetrahydrofolic acid: a potential inhibitor of GAR transformylase and AICAR transformylase, Bioorg. Med. Chem. 2003, 11, 4503–4509.
380. T. H. Marsilje, M. P. Hedrick, J. Desharnais, A. Tavassoli, Y. Zhang, I. A. Wilson, S. J. Benkovic, and D. L. Boger, Design, synthesis, and biological evaluation of simplified a-keto heterocycle, trifluoromethyl ketone, and formyl substituted folate analogues as potential inhibitors of GAR transformylase and AICAR transformylase, Bioorg. Med. Chem. 2003, 11, 4487–4501.
379. P. J. Krenitsky and D. L. Boger, Synthesis of the (S,S,S)-diastereomer of the 15-membered biaryl ring system of RP 66453, Tetrahedron Lett. 2003, 44, 4019–4022.
378. J. P. Parrish, D. Kastrinsky, F. Stauffer, M. P. Hedrick, I. Hwang, and D. L. Boger, Establishment of substituent effects in the DNA binding subunit of CBI analogues of the duocarmycins and CC-1065, Bioorg. Med. Chem. 2003, 11, 3815–3838.
377. Y. Zhang, J. Desharnais, T. H. Marsilje, C. Li, M. P. Hedrick, L. T. Gooljarsingh, A. Tavassoli, S. J. Benkovic, A. J. Olson, D. L. Boger, and I. A. Wilson, Rational design, synthesis, evaluation, and crystal structure of a potent inhibitor of human GAR Tfase: 10-trifluoroacetyl-5,10-dideaza-acylic-5,6,7,8-tetrahydrofolic acid, Biochemistry 2003, 42, 6043–6056.
376. D. R. Soenen, I. Hwang, M. P. Hedrick, and D. L. Boger, Multidrug resistant reversal activity of key ningalin analogues, Bioorg. Med. Chem. Lett. 2003, 13, 1777–1781.
375. D. Leung, C. Hardouin, D. L. Boger, and B. F. Cravatt, Discovering potent and selective reversible inhibitors of enzymes in complex proteomes, Nature Biotechnol. 2003, 21, 687–691.
374. D. R. Soenen, J. M. Zimpleman, and D. L. Boger, Synthesis and inverse electron demand Diels–Alder reactions of 3,6-bis(3,4-dimethoxybenzoyl)-1,2,4,5-tetrazine, J. Org. Chem. 2003, 68, 3593–3598.
373. J. Wanner, D. Tang, C. C. McComas, B. M. Crowley, W. Jiang, and D. L. Boger, A new and improved method for deglycosidation of glycopeptide antibiotics exemplified with vancomycin, ristocetin, and ramoplanin, Bioorg. Med. Chem. Lett. 2003, 13, 1169–1173.
372. D. L. Boger, Solution-phase synthesis of combinatorial libraries designed to modulate protein–protein or protein–DNA interactions, Bioorg. Med. Chem. 2003, 11, 1607–1613. Invited Perspectives article.
371. W. Jiang, J. Wanner, R. J. Lee, P.-Y. Bounaud, and D. L. Boger, Total synthesis of the ramoplanin A2 and ramoplanose aglycon, J. Am. Chem. Soc. 2003, 125, 1877–1887.
370. D. L. Boger and S. L. Castle, Synthesis of cycloisodityrosine peptides, Houben-Weyl, Methods of Organic Chemistry: Synthesis of Peptides and Peptidomimetics; Goodman, M., Ed.; Theime: Stuttgart,2002, 194-206.
369. Y. Zhang, J. Desharnais, S. E. Greasley, G. P. Beardsley, D. L. Boger, and I. A. Wilson, Crystal structures of human GAR Tfase at low and high pH and with substrate β-GAR, Biochemistry 2002, 41, 14206-14215.
368. Y. Ambroise, B. Yuspan, M. H. Ginsberg, and D. L. Boger, Inhibitors of cell migration that inhibit intracellular Paxillin/a4 binding: a well documented use of positional scanning libraries, Chem. Biol. 2002, 9, 1219-1226.
367. S. E. Wolkenberg and Dale L. Boger, Total synthesis of anhydrolycorinone utilizing sequential intramolecular Diels-Alder reactions of a 1,3,4-oxadiazole, J. Org. Chem. 2002, 67, 7361-7364.
366. G. D. Wilkie, G. I. Elliott, B. S. J. Blagg, S. E. Wolkenberg, D. R. Soenen, M. M. Miller, S. Pollack, and D. L. Boger, Intramolecular Diels-Alder and tandem intramolecular Diels-Alder/1,3-dipolar cycloaddition reactions of 1,3,4-oxadiazoles, J. Am. Chem. Soc. 2002, 124, 11292-11294.
365. C. R. Woods, T. Ishii, and D. L. Boger, Synthesis and DNA binding properties of iminodiacetic acid (IDA) linked polyamides: characterization of cooperative extended 2:1 side-by-side parallel binding,J. Am. Chem. Soc. 2002, 124, 10676-10682.
364. C. R. Woods, N. Faucher, B. Eschgfaller, K. W. Bair, and D. L. Boger, Synthesis and DNA binding properties of saturated distamycin analogues, Bioorg. Med. Chem. Lett. 2002, 12, 2647-2650.
363. B. S. J. Blagg and D. L. Boger, Total synthesis of (+)-camptothecin, Tetrahedron 2002, 58, 6343-6349.
362. S. E. Wolkenberg and D. L. Boger, Mechanisms of in situ activation of DNA targeting antitumor agents, Chem. Rev. 2002, 102, 2477-2496.
361. Y. Chen, M. Bilban, C. A. Foster, and D. L. Boger, Solution-phase parallel synthesis of a pharmacophore library of HUN-7293 analogues: a general chemical mutagenesis approach to defining structure-function properties of cyclic (depsi)peptides, J. Am. Chem. Soc. 2002, 124, 5431-5440.
360. W. Jiang, J. Wanner, R. J. Lee, P.-Y. Bounaud, and D. L. Boger, Total synthesis of the ramoplanin A2 and ramoplanose aglycon, J. Am. Chem. Soc. 2002, 124, 5288-5290.
359. T. H. Marsilje, M. A. Labroli, M. P. Hedrick. Q. Jin, J. Desharnais, S. J. Baker, L. J. Gooljarsingh, J. Ramacharan, A. Tavassoli, Y. Zhang, I. A. Wilson, G. P. Bearsley, S. J. Benkovic, and D. L. Boger, 10-Formyl-5,10-dideaza-acyclic-5,6,7,8-tetrahydrofolic acid (10-Formyl-DDACTHF): a potent cytotoxic agent acting by selective inhibition of human GAR Tfase and the de novo purine biosynthetic pathway, Bioorg. Med. Chem. 2002, 10, 2739-2749.
358. J. J. McAtee, S. L. Castle, Q. Jin, and D. L. Boger, Synthesis and evaluation of vancomycin and vancomycin aglycon analogues which bear modifications of the residue 3 asparagine, Bioorg. Med. Chem. Lett. 2002, 12, 1319-1322.
357. T. Berg, S. B. Cohen, J. Desharnais, C. Sonderegger, D. J. Maslyar, J. Goldberg, D. L. Boger, and P. K. Vogt, Small molecule antagonists of Myc/Max dimerization in vitro inhibit Myc induced transformation of chicken embryo fibroblasts, Proc. Natl. Acad. Sci. USA 2002, 99, 3830-3835.
356. C. R. Woods, T. Ishii, B. Wu, K. W. Bair, and D. L. Boger, Hairpin versus extended DNA binding of a substituted β-alanine linked polyamide, J. Am. Chem. Soc. 2002, 124, 2148-2152.
355. J. Goldberg, Q. Jin, Y. Ambroise, S. Satoh, J. Desharnais, K. Capps, and D. L. Boger, Erythropoietin mimetics derived from solution phase combinatorial libraries, J. Am. Chem. Soc. 2002, 124, 544-555.
354. Y. Ambroise and D. L. Boger, The DNA phosphate backbone is not involved in catalysis of the duocarmycin and CC-1065 DNA alkylation reaction, Bioorg. Med. Chem. Lett. 2002, 12, 303-306.
353. P. J. Krenitsky and D. L. Boger, Preparation of the 14-membered L,L-cycloisodityrosine subunit of RP 66453, Tetrahedron Lett. 2002, 43, 407-410.
352. D. Lewy, C.-M. Gauss, and D. L. Boger, Fostriecin: chemistry and biology, Curr. Med. Chem. 2002, 9, 2005-2032.
351. S. E. Greasley, T. H. Marsilje, H. Cai, S. Baker, S. J. Benkovic, D. L. Boger, Unexpected formation of an epoxide-derived multisubstrate adduct inhibitor on the active site of GAR transformylase,Biochemistry 2001, 40, 13538-13547.
350. I. Fedorova, A. Hashimoto, R. A. Fecik, M. P. Hedrick, L. O. Hanus, D. L. Boger, K. C. Rice, and A. S. Basile, Behavioral evidence for the interaction of oleamide with multiple neurotransmitter systems, J. Pharmacol. Exp. Ther. 2001, 299, 332-342.
349. D. L. Boger and W. C. Tse, Thiazole orange as the fluorescent intercalator in a high resolution FID assay for determining DNA binding affinity and sequence selectivity of small molecules, Bioorg. Med. Chem. 2001, 9, 2511-2518.
348. D. L. Boger, H. W. Schmitt, B. E. Fink, and M. P. Hedrick, Parallel synthesis and evaluation of 132 (+)-1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one (CBI) analogues of CC-1065 and the duocarmycins defining the contribution of the DNA-binding domain, J. Org. Chem. 2001, 66, 6654-6661.
347. D. L. Boger, Vancomycin, teicoplanin, and ramoplanin: synthetic and mechanistic studies, Med. Res. Rev. 2001, 21, 356-381.
346. D. A. Ellis, S. E. Wolkenberg, and D. L. Boger, Metal cation complexation and activation of reversed CPyI analogues of CC-1065 and duocarmycin SA: partitioning the effects of binding and catalysis,J. Am. Chem. Soc. 2001, 123, 9299-9306.
345. Y. Mori, J. J. McAtee, O. Rogel, and D. L. Boger, Alternative synthesis and thermal atropisomerism of a fully functionalized DEFG ring system of teicoplanin, Tetrahedron Lett. 2001, 42, 6061-6064.
344. D. L. Boger and J. Hong, Asymmetric total synthesis of ent-(-)-roseophilin: assignment of absolute configuration, J. Am. Chem. Soc. 2001, 123, 8515-8519.
343. D. L. Boger, F. Stauffer, and M. P. Hedrick, Substituent effects within the DNA binding subunit of CBI analogues of the duocarmycins and CC-1065, Bioorg. Med. Chem. Lett. 2001, 9, 2021-2024.
342. D. L. Boger, S. R. Brunette, and R. M. Garbaccio, Synthesis and evaluation of a series of C3-substituted CBI analogues of CC-1065 and the duocarmycins, J. Org. Chem. 2001, 66, 5163-5173.
341. D. L. Boger, H. Miyauchi, and M. P. Hedrick, α-Keto heterocycle inhibitors of fatty acid amide hydrolase: carbonyl group modification and α-substitution, Bioorg. Med. Chem. Lett. 2001, 9, 1517-1520.
340. D. L. Boger, B. E. Fink, S. R. Brunette, W. C. Tse, and M. P. Hedrick, A simple, high resolution method for establishing DNA binding affinity and sequence selectivity, J. Am. Chem. Soc. 2001, 123, 5878-5891.
339. D. L. Boger, S. Ichikawa, and W. Zhong, Total synthesis of fostriecin (CI-920), J. Am. Chem. Soc. 2001, 123, 4161-4167.
338. D. L. Boger, T. V. Hughes, and M. P. Hedrick, Synthesis, chemical properties, and biological evaluation of CC-1065 and duocarmycin analogues incorporating the 5-methoxycarbonyl-1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one alkylation subunit, J. Org. Chem. 2001, 66, 2207-2216.
337. D. L. Boger, S. H. Kim, Y. Mori, J.-H. Weng, O. Rogel, S. L. Castle, and J. J. McAtee, First and second generation total synthesis of the teicoplanin aglycon, J. Am. Chem. Soc. 2001, 123, 1862-1871.
336. D. L. Boger, J. Goldberg, S. Silletti, T. Kessler, and D. A. Cheresh, Identification of a novel class of small-molecule anti-angiogenic agents through screening of combinatorial libraries which function by inhibiting the binding and localization of proteinase MMP2 to integrin αvβ3, J. Am. Chem. Soc. 2001, 123, 1280-1288.
335. D. L. Boger, S. Ichikawa, W. C. Tse, M P. Hedrick, and Q. Jin, Total synthesis of thiocoraline and BE-22179 and assessment of their DNA binding and biological properties, J. Am. Chem. Soc. 2001, 123, 561-568.
334. S. Siletti, T. Kessler, J. Goldberg, D. L. Boger, and D. A. Cheresh, Disruption of MMP2 binding to integrin αvβ3 by a novel organic molecule inhibits angiogenesis and tumor growth in vivo, Proc. Natl. Acad. Sci. USA 2001, 98, 119-124.
333. D. L. Boger and J. Goldberg, Cytokine receptor dimerization and activation: prospects or small molecule agonists, Bioorg. Med. Chem. 2001, 9, 557-562.
332. D. L. Boger, S. Ichikawa, and H. Jiang, Total synthesis of the rubrolone aglycon, J. Am. Chem. Soc. 2000, 122, 12169-122173.
331. D. L. Boger and S. E. Wolkenberg, Total synthesis of Amaryllidaceae alkaloids utilizing sequential intramolecular heterocyclic azadiene Diels-Alder reactions of an unsymmetrical 1,2,4,5-tetrazine, J. Org. Chem. 2000, 65, 9120-9124.
330. D. L. Boger, B. M. Aquila, W. C. Tse, and M. Searcey, Synthesis and evaluation of a novel bleomycin A2 analogue: continuing assessment of the linker domain, Tetrahedron Letters 2000, 41, 9493-9498.
329. D. L. Boger, R. A. Fecik, J. E. Patterson, H. Miyauchi, M. P. Patricelli, and B. F. Cravatt, Fatty acid amide hydrolase substrate specificity, Bioorg. Med. Chem. Lett. 2000, 10, 2613-2616.
328. D. L. Boger, J.-H. Weng, S. Miyazaki, J. J. McAtee, S. L. Castle, S. H. Kim, Y. Mori, O. Rogel, H. Strittmatter, and Q. Jin, Thermal atropisomerism of teicoplanin aglycon derivatives: preparation of the P,P,P and M,P,P atropisomers of the teicoplanin aglycon via selective equilibration of the DE ring system, J. Am. Chem. Soc. 2000, 122, 10047-10055.
327. J. A. Smith, G. Bifulco, D. A. Case, D. L. Boger, L. Gomez-Paloma, and W. J. Chazin, J. Mol. Biol. 2000, 300, 1195-1204.
326. D. L. Boger, R. J. Lee, P.-Y. Bounaud, and P. Meier, Asymmetric synthesis of orthogonally protected L-threo-β-hydroxyasparagine, J. Org. Chem. 2000, 65, 6770-6772.
325. D. L. Boger and J. K. Lee, Development of a solution-phase synthesis of minor groove binding bis-intercalators based on triostin A suitable for combinatorial synthesis, J. Org. Chem. 2000, 65, 5996-6000.
324. D. L. Boger, M. A. Dechantsreiter, T. Ishii, B. E. Fink, and M. P. Hedrick, Assessment of solution phase positional scanning libraries based on distamycin A for the discovery of new DNA binding agents, Bioorg. Med. Chem. 2000, 8, 2049-2057.
323. D. L. Boger, Y. Chen, and C. A. Foster, Synthesis and evaluation of aza HUN-7293, Bioorg. Med. Chem. Lett. 2000, 10, 1741-1744.
322. D. L. Boger, J. Goldberg, S. Satoh, S. B. Cohen, and P. K. Vogt, Non amide-based combinatorial libraries derived from N-BOC iminodiacetic acid: solution-phase synthesis of a 150-membered piperazinone library with activity against LEF-1/β-catenin mediated transcription, Helv. Chim. Acta 2000, 83, 1825-1845.
321. D. L. Boger, S. H. Kim, S. Miyazaki, H. Strittmatter, J.-H. Weng, Y. Mori, O. Rogel, S. L. Castle, and J. J. McAtee, Total synthesis of the teicoplanin aglycon, J. Am. Chem. Soc. 2000, 122, 7416-7417.
320. D. L. Boger, B. E. Fink, and M. P. Hedrick, Total synthesis of distamycin A and 2640 analogs: a solution-phase combinatorial approach to the discovery of new, bioactive DNA binding agents and development of a rapid, high-throughput screen for determining relative DNA binding affinity or DNA binding sequence selectivity, J. Am. Chem. Soc. 2000, 122, 6382-6394.
319. D. L. Boger, S. E. Wolkenberg, and C. W. Boyce, A new method in situ activation for a novel class of DNA alkylating agents: tunable metal cation complexation and activation, J. Am. Chem. Soc. 2000,122, 6325-6326.
318. D. L. Boger, T. H. Marsilje, R. A. Castro, M. P. Hendrick, Q. Jin, S. J. Baker, J. Shim, and S. J. Benkovic, Design, synthesis, and evaluation of fluoronitrophenyl folate analogs as potential GAR transformylase and AICAR transformylase inhibitors, Bioorg. Med. Chem. Lett. 2000, 10, 1471-1475.
317. D. L. Boger and J. Goldberg, Multi-step solution-phase combinatorial chemistry, Chapter 10 in Combinatorial Chemistry: A Practical Approach, Fenniri, H., Ed.; Oxford University Press: Oxford, 2000, 303-326.
316. D. L. Boger, Solution-phase synthesis of combinatorial libraries: to bead or not to bead, in perspective articles on the utility and application of solution-phase combinatorial chemistry, C. M. Baldino, J. Combinatorial Chem. 2000, 2, 89-103.
315. D. L. Boger, A. Santill�n, Jr., M. Searcey, S. R. Brunette, S. E. Wolkenberg, M. P. Hedrick, and Q. Jin, Synthesis and evaluation of 1,2,8,8a-tetrahydrocyclopropo[c]pyrrolo[3,2-e]indol-4(5H)-one, the parent alkylation subunit of CC-1065 and the duocarmycins: impact of the alkylation subunit substituents and its implications for DNA alkylation catalysis, J. Org. Chem. 2000, 65, 4101-4111.
314. D. L. Boger and C. W. Boyce, Selective metal cation activation of a DNA alkylating agent: synthesis and evaluation of methyl 1,2,9,9a-tetrahydrocyclopropa[c]pyrido[3,2-e]indol-4-one-7-carboxylate, J. Org. Chem. 2000, 65, 4088-4100.
313. D. L. Boger, B. E. Fink, and M. P. Hedrick, A new class of highly cytotoxic diketopiperazines, Bioorg. Med. Chem. Lett. 2000, 10, 1019-1020.
312. D. L. Boger, H. Sato, A. E. Lerner, M. P. Hedrick, R. A. Fecik, H. Miyauchi, G. D. Wilkie, B. J. Austin, M. P. Patricelli, and B. F. Cravatt, Exceptionally potent inhibitors of fatty acid amide hydrolase: the enzyme responsible for degradation of endogenous oleamide and anandamide, Proc. Natl. Acad. Sci. USA 2000, 97, 5044-5049.
311. D. L. Boger, D. R. Soenen, C. W. Boyce, M. P. Hedrick, and Q. Jin, Total synthesis of ningalin B utilizing a heterocyclic azadiene Diels-Alder reaction and discovery of a new class of potent multidrug resistant (MDR) reversal agents, J. Org. Chem. 2000, 65, 2479-2483.
310. D. L. Boger and S. Ichikawa, Total syntheses of thiocoraline and BE-22179: establishment of relative and absolute stereochemistry, J. Am. Chem. Soc. 2000, 122, 2956-2957.
309. D. L. Boger, M. A. Labroli, T. H. Marsilje, Q. Jin, M. P. Hedrick, S. J. Baker, J. H. Shim, and S. J. Benkovic, Conformationally restricted analogs designed for selective inhibition of GAR Tfase versus thymidylate synthase or dihydrofolate reductase, Bioorg. Med. Chem. 2000, 8, 1075-1086.
308. D. L. Boger, M. Searcey, W. C. Tse, and Q. Jin, Bifunctional alkylating agents derived from duocarmycin SA: potent antitumor activity with altered sequence selectivity, Bioorg. Med. Chem. Lett. 2000,10, 495-498.
307. D. L. Boger, J. K. Lee, J. Goldberg, and Q. Jin, Two comparisons of the performance of positional scanning and deletion synthesis for the identification of active constituents in mixture combinatorial libraries, J. Org. Chem. 2000, 65, 1467-1474.
306. P. G. Baraldi, B. Cacciari, R. Romagnoli, G. Spalluto, C. W. Boyce, and D. L. Boger, Resolution of a CPzI precursor. Synthesis and biological evaluation of (+)- and (-)-NBoc-CPzI: a further validation of the relationship between chemical solvolytic stability and cytotoxicity, Bioorg. Med. Chem. Lett. 1999, 9, 3087-3092.
305. D. L. Boger and R. M. Garbaccio, Shape-dependent catalysis: insights into the source of catalysis for the CC-1065 and duocarmycin DNA alkylation reaction, Acc. Chem. Res. 1999, 32, 1043-1052.
304. S. M. Greasley, M. M. Yamashita, H. Cai, S. J. Benkovic, D. L. Boger, and I. A. Wilson, New insights into inhibitor design from the crystal structure and NMR studies of E. coli glycinamide ribonucleotide transformylase in complex with β-GAR and the inhibitor 10-formyl-5,8,10-trideazafolic acid, Biochemistry 1999, 38, 16783-16793.
303. D. L. Boger, M. W. Ledeboer, M. Kume, M. Searcey, and Q. Jin, Total synthesis and comparative evaluation of luzopeptin A-C and quinoxapeptin A-C, J. Am. Chem. Soc. 1999, 121, 11375-11383.
302. D. L. Boger, S. Miyazaki, S. H. Kim, J. H. Wu, S. L. Castle, O. Loiseleur, and Q. Jin, Total synthesis of the vancomycin aglycon, J. Am. Chem. Soc. 1999, 121, 10004-10011.
301. D. L. Boger and R. M. Garbaccio, A novel class of CC-1065 and duocarmycin analogs subject to mitomycin related reductive activation, J. Org. Chem. 1999, 64, 8350-8362.
300. D. L. Boger, W. Jiang, and J. Goldberg, Convergent solution phase synthesis of combinatorial libraries through rigid biaryl or diacetylene couplings, J. Org. Chem. 1999, 64, 7094-7100.
299. D. L. Boger, C. W. Boyce, R. M. Garbaccio, M. Searcey, and Q. Jin, CBI prodrug analogs of CC- 1065 and the duocarmycins, Synthesis 1999, 1505-1509. 30th Anniversary Celebration issue, topic: Chemistry and Medicine.
298. D. L. Boger and R. M. Garbaccio, Are the duocarmycin and CC-1065 DNA alkylation reactions acid-catalyzed? Solvolysis pH rate profiles suggest they are not, J. Org. Chem. 1999, 64, 5666-5669.
297. D. L. Boger, A. Santill�n, Jr., M. Searcey, and Q. Jin, Synthesis and evaluation of duocarmycin and CC-1065 analogues containing modifications in the subunit linking amide, J. Org. Chem. 1999, 64, 5241-5244.
296. D. L. Boger, H. Keim, B. Oberhauser, E. P. Schreiner, and C. A. Foster, Total synthesis of HUN-7293, J. Am. Chem. Soc. 1999, 121, 6197-6205.
295. J. R. Schnell, R. R. Ketchem, D. L. Boger, and W. J. Chazin, DNA binding-induced alkylation: insights from the structure of a DNA duplex alkylated by the indole derivative of duocarmycin SA, J. Am. Chem. Soc. 1999, 121, 5645-5652.
294. D. L. Boger, M. W. Ledeboer, M. Kume, and Q. Jin, Total synthesis of quinoxapeptin A-C: establishment of absolute stereochemistry, Angew. Chem , Int. Ed. 1999, 38, 2424-2426.
293. D. L. Boger, H. Sato, A. E. Lerner, X. Guan, and N. B. Gilula, Arachidonic acid amide inhibitors of gap junction cell-cell communication, Bioorg. Med. Chem. Lett. 1999, 9, 1151-1154.
292. D. L. Boger, J. Goldberg, and C.-M. Andersson, Solution phase combinatorial synthesis of biaryl libraries employing heterogeneous conditions for catalysis and isolation with size exclusion chromatography for purification, J. Org. Chem. 1999, 64, 2422-2427.
291. B. J. Wilcox, K. J. Ritenour-Rodgers, A. S. Asser, L. E. Baumgart, M. A. Baumgart, D. L. Boger, J. E. Patterson, J. L. DeBlassio, M. A. deLong, U. Glufke, M. E. Henz, L. King III, K. A. Merkler, J. J. Robleski, J. C. Vederas, and D. J. Merkler, N-Acylglycine amidation: implications for the biosynthesis of fatty acid primary amides, Biochemistry 1999, 38, 3235-3245.
290. D. L. Boger and K. W. Saionz, DNA binding properties of key sandramycin analogs: systematic examination of the intercalation chromophore, Bioorg. Med. Chem. 1999, 7, 315-322.
289. D. L. Boger, H. Sato, A. E. Lerner, B. J. Austin, J. E. Patterson, M. P. Patricelli, and B. F. Cravatt, Trifluoromethyl ketone inhibitors of fatty acid amide hydrolase: a probe of structural and conformational features contributing to inhibition, Bioorg. Med. Chem. Lett. 1999, 9, 265-270.
288. D. L. Boger and H. Cai, Bleomycin: synthetic and mechanistic studies, Angew. Chem., Int. Ed. 1999, 38, 448-476.
287. D. L. Boger, S. Miyazaki, S. H. Kim, J. H. Wu, O. Loiseleur, and S. L. Castle, Diastereoselective total synthesis of the vancomycin aglycon with ordered atropisomer equilibrations, J. Am. Chem. Soc.1999, 121, 3226-3227.
286. D. L. Boger, J. Hong, M. Hikota, and M. Ishida, Total synthesis of phomazarin, J. Am. Chem. Soc. 1999, 121, 2471-2477.
285. D. L. Boger, M. W. Ledeboer, and M. Kume, Total synthesis of luzopeptins A-C, J. Am. Chem. Soc. 1999, 121, 1098-1099.
284. D. L. Boger, C. W. Boyce, M. A. Labroli, C. A. Sehon, and Q. Jin, Total syntheses of ningalin A, lamellarin O, lukianol A, and permethyl storniamide A utilizing heterocyclic azadiene Diels-Alder reactions, J. Am. Chem. Soc. 1999, 121, 54-62.
283. D. L. Boger, S. L. Castle, S. Miyazaki, J. H. Wu, R. T. Beresis, and O. Loiseleur, Vancomycin CD and DE macrocyclization and atropisomerism studies, J. Org. Chem. 1999, 64, 70-80.
282. D. L. Boger and P. Turnbull, Synthesis and evaluation of a carbocyclic analog of the CC-1065 and duocarmycin alkylation subunits: role of the vinylogous amide and implications on DNA alkylation catalysis, J. Org. Chem. 1998, 63, 8004-8011.
281. D. L. Boger, A. Santill�n, Jr., M. Searcey, and Q. Jin, The critical role of the linking amide in CC-1065 and the duocarmycins: implications on the source of DNA alkylation catalysis, J. Am. Chem. Soc.1998, 120, 11554-11557.
280. D. L. Boger, T. M. Ramsey, H. Cai, S. T. Hoehn, and J. Stubbe, Definition of the effect and role of the bleomycin A2 valerate substituents: preorganization of a rigid, compact conformation implicated in sequence selective DNA cleavage, J. Am. Chem. Soc. 1998, 120, 9149-9158.
279. D. L. Boger, T. M. Ramsey, H. Cai, S. T. Hoehn, and J. Stubbe, A systematic evaluation of the bleomycin A2 L-threonine side chain: its role in preorganization of a compact conformation implicated in sequence selective DNA cleavage, J. Am. Chem. Soc. 1998, 120, 9139-9148.
278. D. L. Boger, S. Miyazaki, O. Loiseleur, R. T. Beresis, S. L. Castle, J. H. Wu, and Q. Jin, Thermal atropisomerism of aglucovancomycin derivatives: preparation of (M,M,M)- and (P,M,M)-aglucovancomycin, J. Am. Chem. Soc. 1998, 120, 8920-8926.
277. D. L. Boger, J. Goldberg, W. Jiang, W. Chai, P. Ducray, J. K. Lee, R. S. Ozer, and C.-M. Andersson, Higher order iminodiacetic acid libraries for probing protein-protein interactions, Bioorg. Med. Chem. 1998, 6, 1347-1378. (Symposium-in-Print honoring S. Schreiber receipt of Tetrahedron Prize).
276. D. L. Boger, P. Ducray, W. Chai, W. Jiang, and J. Goldberg, Higher order iminodiacetic acid libraries for probing protein-protein interactions, Bioorg. Med. Chem. Lett. 1998, 8, 2339-2344. (Symposium-in-Print on Combinatorial Chemistry).
275. D. L. Boger and G. Sch�le, Synthesis of acyclic precursors to (3S,4S)-4-hydroxy-2,3,4,5-tetrahydropyridazine-3-carboxylic acid and incorporation into a luzopeptin/quinoxapeptin dipeptide, J. Org. Chem. 1998, 63, 6421-6424.
274. D. L. Boger, R. P. Schaum, and R. M. Garbaccio, Regioselective inverse electron demand Diels-Alder reactions of N-acyl 6-amino-3-methylthio-1,2,4,5-tetrazines, J. Org. Chem. 1998, 63, 6329-6337.
273. D. L. Boger, W. Chai, and Q. Jin, Multistep convergent solution-phase combinatorial synthesis and deletion synthesis deconvolution, J. Am. Chem. Soc. 1998, 120, 7220-7225.
272. D. L. Boger, S. J. Henriksen, and B. F. Cravatt, Oleamide: An endogenous sleep-inducing lipid and prototypical member of a new class of biological signaling molecules, Current Pharmaceutical Design1998, 4, 203-214.
271. D. L. Boger, R. T. Beresis, O. Loiseleur, J. H. Wu, and S. L. Castle, Synthesis of the vancomycin CDE ring system, Bioorg. Med. Chem. Lett. 1998, 8, 721-724.
270. M. P. Patricelli, J. E. Patterson, D. L. Boger, and B. F. Cravatt, An endogenous REM sleep inducing compound is a potent competitive inhibitor of fatty acid amide hydrolase (FAAH), Bioorg. Med. Chem. Lett. 1998, 8, 613-618.
269. D. L. Boger, J. E. Patterson, X. Guan, B. F. Cravatt, R. A. Lerner, and N. B. Gilula, Chemical requirements for inhibition of gap junction communication by the biologically active lipid oleamide, Proc. Natl. Acad. Sci. U.S.A. 1998, 95, 4810-4815.
268. D. L. Boger, J. E. Patterson, and Q. Jin, Structural requirements for 5-HT2A and 5-HT1A receptor potentiation by the biologically active lipid oleamide, Proc. Natl. Acad. Sci. U.S.A. 1998, 95, 4102-4107.
267. D. L. Boger and W. Chai, Solution-phase combinatorial synthesis: convergent multiplication of diversity via the olefin metathesis reaction, Tetrahedron 1998, 54, 3955-3970. (Tetrahedron Symposium-in-Print on solution-phase combinatorial synthesis).
266. D. L. Boger, C. W. Boyce, R. M. Garbaccio, and M. Searcey, Synthesis of CC-1065/duocarmycin analogs via intramolecular aryl radical cyclization of a tethered vinyl chloride, Tetrahedron Letters1998, 39, 2227-2230.
265. D. L. Boger, Heterocyclic and acyclic azadiene Diels-Alder Reactions: total synthesis of nothapodytine B, J. Heterocyclic Chem. 1998, 35, 1003-1011. (Award Lecture, 1997 ISHC Katritzky Award).
264. D. L. Boger, M. J. Kochanny, H. Cai, D. Wyatt, P. A. Kitos, M. S. Warren, J. Ramcharan, L. T. Gooljarsingh, and S. J. Benkovic, Design, synthesis, and evaluation of potential GAR and AICAR inhibitors, Bioorg. Med. Chem. 1998, 6, 643-659.
263. W. Wu, D. E. Vanderwall, S. Teramoto, S. M. Lui, S. T. Hoehn, X.-J. Tang, C. J. Turner, D. L. Boger, J. W. Kozarich, and J. Stubbe, NMR studies of Co deglycobleomycin A2 green and its complex with oligonucleotide d(CCAGGCCTGG)2, J. Am. Chem. Soc. 1998, 120, 2239-2250.
262. D. L. Boger and J. Hong, Total synthesis of nothapodytine B and (-)-mappicine, J. Am. Chem. Soc., 1998, 120, 1218-1222.
261. T. M. Ramsey, H. Cai, S. T. Hoehn, J. W. Kozarich, J. Stubbe, and D. L. Boger, Assessment of the role of the bleomycin A2 pyrimidoblamic acid C4 amino group, J. Am. Chem. Soc. 1998, 120, 53-65.
260. D. L. Boger, J.-H. Chen, K. W. Saionz, and Q. Jin, Synthesis of key sandramycin analogs: systematic examination of the intercalation chromophore, Bioorg. Med. Chem. 1998, 6, 85-102.
259. D. L. Boger, R. M. Garbaccio, and Q. Jin, Synthesis and evaluation of CC-1065 and duocarmycin analogs incorporating the iso-CI and iso-CBI alkylation subunits: impact of relocation of the C-4 carbonyl, J. Org. Chem. 1997, 62, 8875-8891.
258. X. Guan, B. F. Cravatt, G. R. Ehring, J. E. Hall, D. L. Boger, R. A. Lerner, and N. B. Gilula, The sleep-inducing lipid oleamide deconvolutes Gap junction communication and calcium wave transmission in glial cells, J. Cell Biol. 1997, 139, 1785-1792.
257. D. L. Boger, O. Loiseleur, S. L. Castle, R. T. Beresis, and J. H. Wu, Thermal atropisomerism of fully functionalized vancomycin CD, DE, and CDE ring systems, Bioorg. Med. Chem. Lett. 1997, 7, 3199-3202.
256. D. L. Boger and P. Turnbull, Synthesis and evaluation of CC-1065 and duocarmycin analogs incorporating the 1,2,3,4,11,11a-hexahydrocyclopropa[c]naphtho[2,1-b]azepin-6-one (CNA) alkylation subunit: structural features that govern reactivity and reaction regioselectivity, J. Org. Chem. 1997, 62, 5849-5863.
255. D. L. Boger, R. S. Ozer, and C.-M. Andersson, Generation of symmetrical compound libraries by solution-phase combinatorial chemistry, Bioorg. Med. Chem. Lett. 1997, 7, 1903-1908.
254. P. G. Eis, J. A. Smith, J. M. Rydzewski, D. A. Case, D. L. Boger, and W. J. Chazin, High resolution solution structure of a DNA duplex alkylated by the antitumor agent duocarmycin SA, J. Mol. Biol.1997, 272, 237-252.
253. D. L. Boger, N.-E. Haynes, M. S. Warren, J. Ramcharan, P. A. Kitos, and S. J. Benkovic, A multisubstrate analog based on 5,8,10-trideazafolate, Bioorg. Med. Chem. 1997, 5, 1853-1858.
252. D. L. Boger, N.-E. Haynes, M. S. Warren, J. Ramcharan, A. E. Marolewski, P. A. Kitos, and S. J. Benkovic, Abenzyl 10-formyl-trideazafolic acid (abenzyl 10-formyl-TDAF): an effective inhibitor of glycinamide ribonucleotide transformylase, Bioorg. Med. Chem. 1997, 5, 1847-1852.
251. D. L. Boger, N.-E. Haynes, M. S. Warren, J. Ramcharan, P. A. Kitos, and S. J. Benkovic, Functionalized analogs of 5,8,10-trideazafolate: development of an enzyme-assembled tight binding inhibitor of GAR Tfase and an irreversible inhibitor of AICAR Tfase, Bioorg. Med. Chem. 1997, 5, 1839-1846.
250. D. L. Boger, N.-E. Haynes, M. S. Warren, L. T. Gooljarsingh, J. Ramcharan, P. A. Kitos, and S. J. Benkovic, Functionalized analogs of 5,8,10-trideazafolate as potential inhibitors of GAR Tfase or AICAR Tfase, Bioorg. Med. Chem. 1997, 5, 1831-1838.
249. D. L. Boger, N.-E. Haynes, P. A. Kitos, M. S. Warren, J. Ramcharan, A. E. Marolewski, and S. J. Benkovic, 10-Formyl-5,8,10-trideazafolic acid (10-formyl-TDAF): a potent inhibitor of glycinamide ribonucleotide transformylase, Bioorg. Med. Chem. 1997, 5, 1817-1830.
248. D. L. Boger, S. Teramoto, and H. Cai, N-Methyl-L-threonine analogs of deglycobleomycin A2: synthesis and evaluation, Bioorg. Med. Chem. 1997, 5, 1577-1590.
247. D. L. Boger, R. M. Borzilleri, S. Nukui, and R. T. Beresis, Synthesis of the vancomycin CD and DE ring systems, J. Org. Chem. 1997, 62, 4721-4736.
246. S. M. Sakya, K. K.Groskopf, and D. L. Boger, Preparation and inverse electron demand Diels-Alder reactions of 3-methoxy-6-methylthio-1,2,4,5-tetrazine, Tetrahedron Lett. 1997, 38, 3805-3808.
245. D. L. Boger, Applications of free radicals in organic synthesis, Israel J. Chem. 1997, 37, 119-129. (Commemorative issue honoring G. Stork and S. J. Danishefsky receipt of the Wolf Prize in Chemistry).
244. D. L. Boger, B. Bollinger, D. L. Hertzog, D. S. Johnson, H. Cai, P. M�sini, R. M. Garbaccio, Q. Jin, and P. A. Kitos, Reversed and sandwiched analogs of duocarmycin SA: establishment of the origin of the sequence selective alkylation of DNA and new insights into the source of catalysis, J. Am. Chem. Soc. 1997, 119, 4987-4998.
243. D. L. Boger, D. L. Hertzog, B. Bollinger, D. S. Johnson, H. Cai, J. Goldberg, and P. Turnbull, Duocarmycin SA shortened, simplified, and extended agents: a systematic examination of the role of the DNA binding subunit, J. Am. Chem. Soc. 1997, 119, 4977-4986.
242. D. L. Boger and J.-H. Chen, An exceptionally potent analog of sandramycin, Bioorg. Med. Chem. Lett. 1997, 7, 919-922.
241. D. L. Boger, C. W. Boyce, R. M. Garbaccio, and J. A. Goldberg, CC-1065 and the duocarmycins: synthetic studies, Chem. Rev. 1997, 97, 787-828.
240. D. L. Boger, J. Zhou, R. M. Borzilleri, S. Nukui, and S. L. Castle, Synthesis of (9R,12S)- and (9S,12S)-cycloisodityrosine and their N-methyl derivatives J. Org. Chem. 1997, 62, 2054-2069.
239. D. L. Boger, W. Chai, R. S. Ozer, and C.-M. Andersson, Solution-phase combinatorial synthesis via the olefin metathesis reaction, Bioorg. Med. Chem. Lett. 1997, 7, 463-468.
238. D. L. Boger, M. Hikota, and B. M. Lewis, Determination of the relative and absolute stereochemistry of fostriecin (CI-920), J. Org. Chem. 1997, 62, 1748-1753.
237. D. L. Boger, C. W. Boyce, and D. S. Johnson, pH Dependence of the rate of DNA alkylation for (+)-duocarmycin SA and (+)-CCBI-TMI, Bioorg. Med. Chem. Lett. 1997, 7, 233-238.
236. D. L. Boger, J. A. McKie, and C. W. Boyce, Asymmetric synthesis of the CBI alkylation subunit of the CC-1065 and duocarmycin analogs, Synlett 1997, 515-517.
235. D. L. Boger, J. A. McKie, T. Nishi, and T. Ogiku, Total synthesis of (+)-duocarmycin A, epi-(+)-duocarmycin A and their unnatural enantiomers: assessment of chemical and biological properties, J. Am. Chem. Soc. 1997, 119, 311-325.
234. D. L. Boger and R. M. Garbaccio, Catalysis of the CC-1065 and duocarmycin DNA alkylation reaction: DNA binding induced conformational change in the agent results in activation, Bioorg. Med. Chem. 1997, 5, 263-276.
233. D. L. Boger and N. Han, CC-1065/duocarmycin and bleomycin A2 hybrid agents: lack of enhancement of DNA alkylation by attachment to noncomplementary DNA binding subunits, Bioorg. Med. Chem. 1997, 5, 233-244.
232. B. F. Cravatt, D. K. Giang, S. P. Mayfield, D. L. Boger, R. A. Lerner, and N. B. Gilula, Molecular characterization of an enzyme that degrades neuromodulatory fatty acid amides, Nature 1996, 384, 83-87.
231. D. L. Boger, B. Bollinger, and D. S. Johnson, Examination of the role of the duocarmycin SA methoxy substituents: identification of the minimum, fully potent DNA binding subunit, Bioorg. Med. Chem. Lett. 1996, 6, 2207-2212.
230. D. L. Boger, J. A. Goldberg, and J. A. McKie, A comparative study of the solvolysis reactivity, regioselectivity, and stereochemistry of the duocarmycin A and SA alkylation subunits, Bioorg. Med. Chem. Lett. 1996, 6, 1955-1960.
229. D. L. Boger and T. J. Jenkins, Synthesis, X-ray structure, and properties of fluorocyclopropane analogs of the duocarmycins incorporating the 9,9-difluoro-1,2,9,9a-tetrahydrocyclopropa-[c]benz[e]indol-4-one (F2CBI) alkylation subunit, J. Am. Chem. Soc. 1996, 118, 8860-8870.
228. D. L. Boger and J. Zhou, Key Analogs of the tetrapeptide subunit of RA-VII and deoxybouvardin, Bioorg. Med. Chem. 1996, 4, 1597-1603.
227. C. M. Tarby, S. Cheng, and D. L. Boger, Solution phase strategy for the synthesis of chemical libraries containing small organic molecules: a general depeptide mimetic and a flexible universal template, in Molecular Diversity and Combinatorial Chemistry: Libraries and Drug Discovery; Chaiken, I. M.; Janda, K. D., Eds.; ACS: Washington; 1996, 81-98.
226. D. L. Boger, N. Han, C. M. Tarby, C. W. Boyce, H. Cai, Q. Jin, and P. A. Kitos, Synthesis, chemical properties, and preliminary evaluation of substituted CBI analogs of CC-1065 and the duocarmycins incorporating the 7-cyano-1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one (CCBI) alkylation subunit: Hammett quantitation of the magnitude of electronic effects on functional reactivity, J. Org. Chem. 1996, 61, 4894-4912.
225. J. E. Patterson, I. R. Ollmann, B. F. Cravatt, Dale L. Boger, C.-H. Wong, and R. A. Lerner, Inhibition of oleamide hydrolase catalyzed hydrolysis of the endogenous sleep-inducing lipid: cis-9-octadecenamide, J. Am. Chem. Soc. 1996, 118, 5938-5945.
224. D. L. Boger, J. Zhou, R. M. Borzilleri, and S. Nukui, Synthesis of (9S,12S)-cycloisodityrosine and its unnatural (9R,12S)-diastereomer, Bioorg. Med. Chem. Lett. 1996, 6, 1089-1092.
223. D. L. Boger and J. Zhou, An alternative synthesis of the cycloisodityrosine subunit of deoxybouvardin and RA-VII and related agents: reassignment of the stereochemistry of prior intermediates, J. Org. Chem. 1996, 61, 3938-3939.
222. D. L. Boger, J. Zhou, and H. Cai, Demonstration and definition of the noncovalent binding selectivity of agents related to CC-1065 by an affinity cleavage agent: noncovalent binding coincidental with alkylation, Bioorg. Med. Chem. 1996, 4, 859-868.
221. S. Cheng, C. M. Tarby, D. D. Comer, J. P. Williams, L. H. Caporale, P. L. Myers, and D. L. Boger, A solution-phase strategy for the synthesis of combinatorial libraries containing small molecules: a universal and dipeptidomimetic template, Bioorg. Med. Chem. 1996, 4, 727-737.
220. D. L. Boger, Cycloaddition reactions of azadienes, cyclopropenone ketals and related systems: scope and applications, Chemtracts: Organic Chem. 1996, 9, 149-189.
219. D. S. Johnson and D. L. Boger, DNA Binding Agents, Chapter 3 in Comprehensive Supramolecular Chemistry, Vol. 4, J.-M. Lehn, Series Ed., Y. Murakami, Vol. Ed., Pergamon Press, Oxford, England, 1996, 73-176.
218. D. L. Boger, Azadiene Diels-Alder reactions: scope and applications. Total synthesis of natural and ent-fredericamycin A, J. Heterocycl. Chem. 1996, 33, 1519-1531. (review)
217. D. L. Boger and D. S. Johnson, CC-1065 and the duocarmycins: understanding their biological function through mechanistic studies, Angew. Chem., Int. Ed. Engl. 1996, 35, 1438-1474.
216. D. L. Boger, T. M. Ramsey, and H. Cai, Synthesis and Evaluation of potential Nπ and Nσ metal chelation sites within the β-hydroxy-L-histidine subunit of bleomycin A2: functional characterization of imidazole Nπ metal complexation, Bioorg. Med. Chem. 1996, 4, 195-208.
215. D. L. Boger, S. Teramoto, and H. Cai, Synthesis and evaluation deglycobleomycin A2 analogs containing a tertiary N-methyl amide and simple ester replacement for the L-histidine secondary amide: direct functional characterization of the requirement for secondary amide metal complexation, Bioorg. Med. Chem. 1996, 4, 179-194.
214. D. L. Boger, R. M. Borzilleri, and S. Nukui, Synthesis of (R)-3,5-dihydroxy-4-methoxyphenylglycine derivatives: the central amino acid of vancomycin and related agents, J. Org. Chem. 1996, 61, 3561-3565.
213. D. L. Boger, J. A. McKie, N. Han, C. M. Tarby, H. W. Riggs, and P. A. Kitos, A Hammett correlation for CC-1065 and duocarmycin analogs: magnitude of substituent electronic effects on functional reactivity Bioorg. Med. Chem. Lett. 1996, 6, 659-664.
212. D. L. Boger, J. A. McKie, H. Cai, B. Cacciari, and P. G. Baraldi, Synthesis and properties of substituted CBI analogs of CC-1065 and the duocarmycins incorporating the 7-methoxy-1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one (MCBI) alkylation subunit: magnitude of electronic effects on the functional reactivity, J. Org. Chem. 1996, 61, 1710-1729.
211. S. Cheng, D. D. Comer, J. P. Williams, P. L. Myers, and D. L. Boger, A novel solution-phase strategy for the synthesis of chemical libraries containing small organic molecules, J. Am. Chem. Soc. 1996,118, 2567-2573.
210. D. L. Boger, J. A. McKie, T. Nishi, and T. Ogiku, Enantioselective total synthesis of (+)-duocarmycin A, epi-(+)-duocarmycin A and their unnatural enantiomers, J. Am. Chem. Soc. 1996, 118, 2301-2302.
209. D. L. Boger, C. M. Tarby, P. L. Myers, and L. H. Caporale, A generalized dipeptidomimetic template: solution phase parallel synthesis of combinatorial libraries, J. Am. Chem. Soc. 1996, 118, 2109-2110.
208. D. L. Boger, J.-H. Chen, and Kurt W. Saionz, (-)-Sandramycin: total synthesis and characterization of DNA binding properties, J. Am. Chem. Soc. 1996, 118, 1629-1644.
207. B. F. Cravatt, R. A. Lerner, and D. L. Boger, Structure determination of an endogenous sleep-inducing lipid, cis-9-octadecenoamide (oleamide): a synthetic approach to the chemical analysis of trace quantities of a natural product, J. Am. Chem. Soc. 1996, 118, 580-590.
206. D. L. Boger, R. M. Borzilleri, and S. Nukui, Synthesis of appropriately functionalized vancomycin CD and DE ring systems, Bioorg. Med. Chem. Lett. 1995, 5, 3091-3096.
205. D. L. Boger and K. Takahashi, Total synthesis of granditropone, grandirubrine, imerubrine and isoimerubrine, J. Am. Chem. Soc. 1995, 117, 12452-12459.
204. D. L. Boger, O. H�ter, K. Mbiya, and M. Zhang, Total synthesis of natural and ent-fredericamycin, J. Am. Chem. Soc. 1995, 117, 11839-11849.
203. D. L. Boger and P. Mesini, DNA alkylation properties of CC-1065 and duocarmycin analogs incorporating the 2,3,10,10a-tetrahydrocyclopropa[d]benzo[f]quinol-5-one (CBQ) alkylation subunit: identification of subtle structural features that contribute to the regioselectivity of the adenine N3 alkylation reaction, J. Am. Chem. Soc. 1995, 117, 11647-11655.
202. D. L. Boger, J. Zhou, B. Winter, and P. A. Kitos, Key analogs of the tetrapeptide subunit of RA-VII and deoxybouvardin, Bioorg. Med. Chem. 1995, 3, 1579-1593.
201. D. L. Boger, W. Yun, and N. Han, 1,2,9,9a-Tetrahydrocyclopropa[c]benz[e]indol-4-one (CBI) analogs of CC-1065 and the duocarmycins: synthesis and evaluation, Bioorg. Med. Chem. 1995, 3, 1429-1453.
200. D. L. Boger and J.-H. Chen, A modified Friedlander condensation for the synthesis of 3-hydroxyquinoline-2-carboxylates, J. Org. Chem. 1995, 60, 7369-7371.
199. D. L. Boger, S. L. Colletti, S. Teramoto, T. M. Ramsey, and J. Zhou, Synthesis of key analogs of bleomycin A2 that permit a systematic evaluation of the linker region: identification of an exceptionally prominent role for the L-threonine substituent, Bioorg. Med. Chem. 1995, 3, 1281-1295.
198. D. L. Boger and J. Zhou, N-Desmethyl derivatives of deoxybouvardin and RA-VII: synthesis and evaluation, J. Am. Chem. Soc. 1995, 117, 7364-7378.
197. D. L. Boger, M. A. Patane, and J. Zhou, Solution phase conformational studies of the cyclic peptide RA-VII: lithium chloride perturbation of the conformational equilibria, J. Am. Chem. Soc. 1995, 117, 7357-7363.
196. D. L. Boger, S. Teramoto, and J. Zhou, Key synthetic analogs of bleomycin A2 that directly address the effect and role of the disaccharide: demannosylbleomycin A2 and a-D-mannopyranosyldeglycobleomycin A2, J. Am. Chem. Soc. 1995, 117, 7344-7356.
195. D. L. Boger, S. Teramoto, T. Honda, and J. Zhou, synthesis and evaluation of the fully functionalized bleomycin A2 metal binding domain containing the 2-O-(3-O-carbamoyl-D-mannopyranosyl)-L-gulopyranosyl disaccharide, J. Am. Chem. Soc. 1995, 117, 7338-7343.
194. D. L. Boger and R. M. Borzilleri, An unusually facile SNAr 14-membered ring biaryl ether macrocyclization reaction suitable for preparation of the cycloisodityrosine subunit of bouvardin, deoxybouvardin and related agents, Bioorg. Med. Chem. Lett. 1995, 5, 1187-1190.
193. B. F. Cravatt, O. Prospero-Garcia, G. Szuidak, S. Henriksen, D. L. Boger, and R. A. Lerner, Chemical characterization of a family of brain lipids that induce sleep, Science 1995, 268, 1506-1509.
192. D. L. Boger and D. S. Johnson, CC-1065 and the duocarmycins: unraveling the keys to a new class of naturally derived DNA alkylating agents, Proc. Natl. Acad. Sci. U.S.A. 1995, 92, 3642-3649.
191. D. L. Boger, W. Yun, H. Cai, and N. Han, CBI-CDPBO1 and CBI-CDPBI1: CC-1065 analogs containing deep-seated modifications in the DNA binding subunits, Bioorg. Med. Chem. 1995, 3, 761-775.
190. D. L. Boger, W. Yun, N. Han, and D. S. Johnson, CC-1065 CBI analogs: an example of DNA alkylation efficiency enhancement through introduction of stabilizing electrostatic interactions, Bioorg. Med. Chem. 1995, 3, 611-621.
189. D. L. Boger and J. A. McKie, An efficient synthesis of 1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one (CBI): an enhanced and simplified analog of the CC-1065 and duocarmycin alkylation subunits, J. Org. Chem. 1995, 60, 1271-1275.
188. D. L. Boger and D. S. Johnson, Second definitive test of proposed models for the origin of the CC-1065 and duocarmycin DNA alkylation selectivity, J. Am. Chem. Soc. 1995, 117, 1443-1444.
187. D. L. Boger, The duocarmycins: synthetic and mechanistic studies, Acc. Chem. Res. 1995, 28, 20-29.
186. D. L. Boger and T. Nishi, Diastereoselective Dieckmann condensation suitable for introduction of the duocarmycin A C6 center: development of a divergent strategy for the total synthesis of duocarmycins A and SA, Bioorg. Med. Chem. 1995, 3, 67-77.
185. D. L. Boger, Total synthesis of (+)- and ent-(-)-duocarmycin SA, in Advances in Nitrogen Heterocycles, Vol. 1, C. J. Moody, Ed., JAI Press, 1995, 229-247.
184. D. L. Boger and P. Mesini, Design, synthesis and evaluation of CC-1065 and duocarmycin analogs incorporating the 2,3,10,10a-tetrahydro-1H-cyclopropa[d]benzo[f]quinol-5-one (CBQ) alkylation subunit: identification and structural origin of subtle stereoelectronic features that govern reactivity and regioselectivity, J. Am. Chem. Soc. 1994, 116, 11335-11348.
183. D. L. Boger, M. A. Patane, and J. Zhou, Total synthesis of bouvardin, O-methyl bouvardin and O-methyl N9-desmethylbouvardin, J. Am. Chem. Soc. 1994, 116, 8544-8556.
182. R. A. Lerner, G. Siuzdak, O. Prospero-Garcia, S. J. Hendriksen, Dale L. Boger, and B. F. Cravatt, Cerebrodiene: a brain lipid isolated from sleep-deprived cats, Proc. Natl. Acad. Sci. U.S.A. 1994, 91, 9505-9508.
181. D. L. Boger, R. A. Lerner, and B. F. Cravatt, Synthesis of a functionalized rigid bicyclo[2.2.1]heptane: a useful hapten for eliciting catalytic antibodies, J. Org. Chem. 1994, 59, 5078-5079.
180. D. L. Boger and M. J. Kochanny, Inverse electron demand Diels-Alder reactions of heterocyclic azadienes: [4 + 2] cycloaddition reactions of amidines with 1,3,5-triazines, J. Org. Chem. 1994, 59, 4950-4955.
179. D. L. Boger, T. Nishi, and B. R. Teegarden, A p-quinonemethide analog of the CC-1065 and duocarmycin alkylation subunits, J. Org. Chem. 1994, 59, 4943-4949.
178. D. L. Boger and Y. Zhu, Diels-Alder reactions of cyclopropenone ketals: a concise tropolone annulation applicable to rubrolone C ring introduction, J. Org. Chem. 1994, 59, 3453-3458.
177. D. L. Boger and W. Yun, CBI-TMI: synthesis and evaluation of a key analog of the duocarmycins. Validation of a direct relationship between chemical solvolytic stability and cytotoxic potency and confirmation of the structural features responsible for the distinguishing behavior of enantiomeric pairs of agents, J. Am. Chem. Soc. 1994, 116, 7996-8006.
176. D. L. Boger, P. Mesini, and C. M. Tarby, Chemical and structural comparison of N-BOC-CBQ and N-BOC-CBI: identification and structural origin of an unappreciated but productive stability of the CC-1065 and duocarmycin SA alkylation subunits, J. Am. Chem. Soc. 1994, 116, 6461-6462.
175. B. F. Cravatt, J. A. Ashley, K. D. Janda, D. L. Boger, and R. A. Lerner, Crossing extreme mechanistic barriers by antibody catalysis: syn elimination to a cis olefin, J. Am. Chem. Soc. 1994, 116, 6013-6014.
174. D. L. Boger and T. Honda, Total synthesis of bleomycin A2 and related agents. 4. Synthesis of the disaccharide subunit: 2-O-(3-O-carbamoyl--D-mannopyranosyl)-L-gulopyranose and completion of the total synthesis of bleomycin A2, J. Am. Chem. Soc. 1994, 116, 5647-5656.
173. D. L. Boger, T. Honda, R. F. Menezes, and S. L. Colletti, Total synthesis of bleomycin A2 and related agents. 3. Synthesis and comparative evaluation of deglycobleomycin A2, epi-deglycobleomycin A2, deglycobleomycin A1, desacetamido, descarboxamido, desmethyl and desimidazole deglycobleomycin A2, J. Am. Chem. Soc. 1994, 116, 5631-5646.
172. D. L. Boger, T. Honda, and Q. Dang, Total synthesis of bleomycin A2 and related agents. 2. Synthesis of (-)-pyrimidoblamic acid, epi-(+)-pyrimidoblamic acid, (+)-desacetamidopyrimidoblamic acid, and (-)-descarboxamidopyrimidoblamic acid, J. Am. Chem. Soc. 1994, 116, 5619-5630.
171. D. L. Boger, S. L. Colletti, T. Honda, and R. F. Menezes, Total synthesis of bleomycin A2 and related agents. 1. Synthesis and DNA binding properties of the extended C-terminus: tripeptide S, tetrapeptide S, pentapeptide S, and related agents, J. Am. Chem. Soc. 1994, 116, 5607-5618.
170. D. L. Boger and W. Yun, Role of the CC-1065 and duocarmycin N2 substituent: validation of a direct relationship between solvolysis chemical stability and in vitro biological potency, J. Am. Chem. Soc.1994, 116, 5523-5524.
169. D. L. Boger, Design, synthesis, and evaluation of DNA minor groove binding agents: the duocarmycins, Pure Appl. Chem. 1994, 66, 837-844.
168. F. Mohamadi, M. M. Spees, G. S. Staten, P. Marder, J. K. Kipka, D. A. Johnson, D. L. Boger, and H. Zarrinmayeh, Total synthesis and biological properties of novel antineoplastic (chloromethyl)furanoindolines: an asymmetric hydroboration mediated synthesis of the alkylation subunits, J. Med. Chem. 1994, 37, 232-239.
167. W. Wrasidlo, D. S. Johnson, and D. L. Boger, Induction of endonucleolytic DNA fragmentation and apoptosis by the duocarmycins, Bioorg. Med. Chem. Lett. 1994, 4, 631-636.
166. D. L. Boger, D. S. Johnson, W. Yun, and C. M. Tarby, Molecular basis for sequence selective DNA alkylation by (+)- and ent-(-)-CC-1065 and related agents: alkylation site models that accommodate the offset AT-rich adenine N3 alkylation selectivity, Bioorg. Med. Chem. 1994, 2, 115-135.
165. D. L. Boger, M. A. Patane, Q. Jin, and P. A. Kitos, Design, synthesis and evaluation of bouvardin, deoxybouvardin and RA-I - XIV pharmacophore analogs, Bioorg. Med. Chem. 1994, 2, 85-100.
164. D. L. Boger, D. S. Johnson, and W. Yun, (+)- and ent-(-)-Duocarmycin SA and (+)- and ent-(-)-N-BOC-DSA DNA alkylation properties. Alkylation site models that accommodate the offset AT-rich adenine N3 alkylation selectivities of the enantiomeric agents, J. Am. Chem. Soc. 1994, 116, 1635-1656.
163. D. L. Boger, T. Honda, R. F. Menezes, S. L. Colletti, Q. Dang, and W. Yang, Total synthesis of (+)-P-3A, epi-(-)-P-3A, and (-)-desacetamido P-3A, J. Am. Chem. Soc. 1994, 116, 82-92.
162. D. L. Boger and J.-H. Chen, (-)-Sandramycin: total synthesis and preliminary DNA binding properties, J. Am. Chem. Soc. 1993, 115, 11624-11625.
161. D. L. Boger and J. Zhou, Total synthesis of (+)-piperazinomycin, J. Am. Chem. Soc. 1993, 115, 11426-11433.
160. D. L. Boger and C. M. Baldino, d,l- and meso-Isochrysohermidin: total synthesis and interstrand DNA cross-linking, J. Am. Chem. Soc. 1993, 115, 11418-11425.
159. D. L. Boger, K. C. Cassidy, and S. Nakahara, Total synthesis of streptonigrone, J. Am. Chem. Soc. 1993, 115, 10733-10741.
158. D. L. Boger and W. Yun, Reversibility of the duocarmycin A and SA DNA alkylation reaction, J. Am. Chem. Soc. 1993, 115, 9872-9873.
157. D. L. Boger, K. Machiya, D. L. Hertzog, P. A. Kitos, and D. Holmes, Total Synthesis and preliminary evaluation of (+)- and ent-(-)-duocarmycin SA, J. Am. Chem. Soc. 1993, 115, 9025-9036.
156. H. H. Wasserman, R. W. DeSimone, D. L. Boger, and C. M. Baldino, Singlet oxygen oxidation of bipyrroles: total synthesis of d,l- and meso-isochrysohermidin, J. Am. Chem. Soc. 1993, 115, 8457-8458.
155. D. L. Boger and J. Zhou, CDPI3-enediyne and CDPI3-EDTA conjugates: a new class of DNA cleaving agents, J. Org. Chem. 1993, 58, 3018-3024.
154. D. L. Boger, D. Yohannes, J. Zhou, and M. A. Patane, Total synthesis of cycloisodityrosine, RA-VII, deoxybouvardin, and N29-desmethyl RA-VII: identification of the pharmacophore and reversal of the subunit functional roles, J. Am. Chem. Soc. 1993, 115, 3420-3430.
153. D. L. Boger and W. L. Corbett, 4p participation of 1-aza-1,3-butadienes in [4 + 2] cycloaddition reactions: intramolecular Diels-Alder reactions of a,b-unsaturated N-sulfonylimines, J. Org. Chem. 1993,58, 2068-2074.
152. D. L. Boger and T. Honda, Studies on the synthesis of bleomycin A2 and P-3A: a diastereoselective imine addition reaction for C2-acetamido side chain introduction, Tetrahedron Letters 1993, 34, 1567-1570.
151. D. L. Boger, D. S. Johnson, M. S. S. Palanki, P. A. Kitos, J. Chang, and P. Dowell, Evaluation of functional analogs of CC-1065 and the duocarmycins incorporating the cross-linking 9a-chloromethyl-1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one (C2BI) alkylation subunit, Bioorg. Med. Chem. 1993, 1, 27-38.
150. D. L. Boger, Y. Nomoto, and B. R. Teegarden, Vancomycin and ristocetin models: synthesis via the Ullmann macrocyclization reaction, J. Org. Chem. 1993, 58, 1425-1433.
149. D. L. Boger, Design, synthesis and evaluation of DNA minor groove binding agents, Pure Appl. Chem. 1993, 65, 1123-1132.
148. D. L. Boger, R. F. Menezes, and T. Honda, Total synthesis of (-)-pyrimidoblamic acid and deglycobleomycin A2, Angew. Chem., Int. Ed. Engl. 1993, 32, 273-275.
147. D. L. Boger and D. Yohannes, K-13 and OF4949: evaluation of key partial structures and pharmacophore delineation, Bioorg. Med. Chem. Lett. 1993, 3, 245-250.
146. D. L. Boger and W. Yang, P-3A and (-)-desacetamido P-3A: demonstration and study of their effective functional cleavage of duplex DNA, Bioorg. Med. Chem. Lett. 1992, 2, 1649-1654.
145. D. L. Boger and K. Machiya, Total synthesis of (+)-duocarmycin SA, J. Am. Chem. Soc. 1992, 114, 10056-10058.
144. D. L. Boger and M. S. S. Palanki, Functional analogs of CC-1065 and the duocarmycins incorporating the 9a-chloromethyl-1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one (C2BI) alkylation subunit: synthesis and preliminary DNA alkylation studies, J. Am. Chem. Soc. 1992, 114, 9318-9327.
143. D. L. Boger, R. F. Menezes, and W. Yang, A simple procedure for the purification and isolation of bleomycin A2, Bioorg. Med. Chem. Lett. 1992, 2, 959-962.
142. D. L. Boger and W. L. Corbett, A convenient and general preparation of N-sulfonylimines, J. Org. Chem. 1992, 57, 4777-4780.
141. D. L. Boger, W. Yun, S. Terashima, Y. Fukuda, K. Nakatani, P. A. Kitos and Q. Jin, DNA alkylation properties of the duocarmycins: (+)-duocarmycin A, epi-(+)-duocarmycin A, ent-(-)-duocarmycin A, and epi,ent-(-)-duocarmycin A, Bioorg. Med. Chem. Lett. 1992, 2, 759-765.
140. D. L. Boger, R. F. Menezes, and Q. Dang, Synthesis of desacetamidopyrimidoblamic acid and deglyco desacetamidobleomycin A2, J. Org. Chem. 1992, 57, 4333-4336.
Correction: J. Org. Chem. 1992, 57, 5276.
139. D. L. Boger and R. F. Menezes, Synthesis of tri- and tetrapeptide S: the extended C-terminus of bleomycin A2, J. Org. Chem. 1992, 57, 4331-4333.
138. D. L. Boger and M. Zhang, A concise synthesis of the fredericamycin A DEF ring system: [4 + 2] cycloaddition reactions of 1-aza-1,3-butadienes, J. Org. Chem. 1992, 57, 3974-3977.
137. D. L. Boger, W. Yun, and B. R. Teegarden, An improved synthesis of 1,2,9,9a-tetrahydrocyclopropa[c]benz[e]indol-4-one (CBI): a simplified analog of the CC-1065 alkylation subunit, J. Org. Chem.1992, 57, 2873-2876.
136. D. L. Boger and S. A. Munk, DNA alkylation properties of CBI-based agents: enhanced functional analogs of CC-1065 incorporating the 1,2,9,9a-tetrahydrocycloprop[1,2-c]benz[1,2-e]indol-4-one (CBI) alkylation subunit, J. Am. Chem. Soc. 1992, 114, 5487-5496.
135. D. L. Boger, R. F. Menezes, Q. Dang, and W. Yang, Deglyco GABA,gly-desacetamidobleomycin A2: a simplified synthetic model for bleomycin A2, Bioorg. Med. Chem. Lett. 1992, 2, 261-266.
134. D. L. Boger and T. T. Curran, Synthesis of the lower subunit of rhizoxin, J. Org. Chem. 1992, 57, 2235-2244.
133. D. L. Boger and Q. Dang, Synthesis of desacetamido P-3A: 1,3,5-triazine-pyrimidine heteroaromatic azadiene Diels-Alder reaction, J. Org. Chem. 1992, 57, 1631-1633.
132. D. L. Boger and R. J. Mathvink, Acyl radicals: intermolecular and intramolecular alkene addition reactions, J. Org. Chem. 1992, 57, 1429-1443.
131. D. L. Boger, D. Yohannes, and J. B. Myers, Jr., Synthesis of N29-desmethyl RA-VII. Identification of the pharmacophore of RA-I - RA-VII and deoxybouvardin and reassignment of the subunit functional roles, J. Org. Chem. 1992, 57, 1319-1321.
130. D. L. Boger and S. M. Sakya, CC-1065 partial structures: enhancement of the noncovalent affinity for DNA minor groove binding through introduction of stabilizing electrostatic interactions, J. Org. Chem. 1992, 57, 1277-1284.
129. D. L. Boger, Design, synthesis, and evaluation of functional analogs of (+)-CC-1065 and the duocarmycins, Proceedings of the 35th Robert A. Welch Foundation Conference on Chemical Research, Chemistry at the Frontiers of Medicine 1992, 35, 137-179.
128. D. L. Boger, Design, synthesis, and evaluation of functional analogs of CC-1065 and the duocarmycins: a detailed study of factors effecting DNA binding selectivity, in Advances in Heterocyclic Natural Products Synthesis, Vol. 2, W. H. Pearson, ed., JAI Press, 1992, 1-188.
127. D. L. Boger and Y. Zhu, Studies on the synthesis of rubrolone: 4p participation of O-alkyl a,b-unsaturated oximes in intramolecular [4 + 2] cycloaddition reactions, Tetrahedron Letters 1991, 32, 7643-7646.
126. D. L. Boger, Duocarmycins: a new class of sequence selective DNA minor groove alkylating agents, Chemtracts: Organic Chem. 1991, 4, 329-349.
125. D. L. Boger, [4 + 2] Cycloadditions: Heterodiene Additions, Chapter 4.3 in Comprehensive Organic Synthesis, Vol. 5, B. M. Trost and I. Fleming, Series Ed.; L. Paquette, Vol. Ed., Pergamon Press, Oxford, England, 1991, 451-512.
124. D. L. Boger and C. M. Baldino, Singlet oxygen mediated oxidative decarboxylation of pyrrole-2-carboxylic acids, J. Org. Chem. 1991, 56, 6942-6944.
123. D. L. Boger and J. B. Myers, Jr., Design and synthesis of a conformational analog of deoxybouvardin, J. Org. Chem. 1991, 56, 5385-5390.
122. D. L. Boger, J. B. Myers, Jr., D. Yohannes, P. A. Kitos, O. Suntornwat, and J. C. Kitos, Evaluation of bouvardin, deoxybouvardin, and RA-I - RA-VII partial structures: reassignment of the pharmacophore, Bioorg. Med. Chem. Lett. 1991, 1, 313-316.
121. D. L. Boger, J. S. Panek, and M. Patel, Preparation and inverse electron demand Diels-Alder reaction of an electron-deficient heterocyclic azadiene: dimethyl 1,2,4,5-tetrazine-3,6-dicarboxylate. 1,2-Diazine and pyrrole introduction, Organic Syntheses 1991, 70, 79-92.
120. D. L. Boger, S. M. Sakya, and D. Yohannes, Total synthesis of combretastatin D-2: intramolecular Ullmann macrocyclization reaction, J. Org. Chem. 1991, 56, 4204-4207.
119. D. L. Boger, T. Ishizaki, and H. Zarrinmayeh, Isolation and characterization of the duocarmycin-adenine DNA adduct, J. Am. Chem. Soc. 1991, 113, 6645-6649.
118. D. L. Boger and M. Zhang, Total synthesis of (±)-cis- and trans-trikentrin A: Diels-Alder reactions of heteroaromatic azadienes, J. Am. Chem. Soc. 1991, 113, 4230-4234.
117. D. L. Boger, S. A. Munk, and H. Zarrinmayeh, The (+)-CC-1065 DNA alkylation: key studies demonstrating a noncovalent binding selectivity contribution to the alkylation selectivity, J. Am. Chem. Soc.1991, 113, 3980-3983.
116. D. L. Boger, S. A. Munk, and T. Ishizaki, The (+)-CC-1065 DNA alkylation: observation of an unexpected relationship between cyclopropane electrophile reactivity and the intensity of DNA alkylation,J. Am. Chem. Soc. 1991, 113, 2779-2780.
115. D. L. Boger and I. C. Jacobson, Synthesis and preliminary evaluation of the fredericamycin A ABCDE ring system, J. Org. Chem. 1991, 56, 2115-2122.
114. D. L. Boger, T. Ishizaki, S. M. Sakya, S. A. Munk, P. A. Kitos, Q. Jin, and J. M. Besterman, Synthesis and preliminary evaluation of (+)-CBI-indole2: an enhanced functional analog of (+)-CC-1065,Bioorg. Med. Chem. Lett. 1991, 1, 115-120.
113. D. L. Boger, S. A. Munk, H. Zarrinmayeh, T. Ishizaki, J. Haught, and M. Bina, An alternative and convenient strategy for generation of substantial quantities of singly 5'-32P-end-labeled double-stranded DNA for binding studies: development of a protocol for the examination of the functional features of (+)-CC-1065 and the duocarmycins that contribute to their sequence-selective DNA binding properties,Tetrahedron 1991, 47, 2661-2682.
112. D. L. Boger and D. Yohannes, Intramolecular Ullmann condensation reaction: an effective approach to macrocyclic diaryl ethers, J. Org. Chem. 1991, 56, 1763-1767.
111. D. L. Boger, W. L. Corbett, T. T. Curran, and A. M. Kasper, Inverse electron demand Diels-Alder reactions of N-sulfonyl a,b-unsaturated imines: a general approach to the implementation of the 4pparticipation of 1-aza-1,3-butadienes in Diels-Alder reactions, J. Am. Chem. Soc. 1991, 113, 1713-1729.
110. D. L. Boger, H. Zarrinmayeh, S. A. Munk, P. A. Kitos, and O. Suntornwat, Demonstration of a pronounced effect of noncovalent binding selectivity on the (+)-CC-1065 DNA alkylation and identification of the pharmacophore of the alkylation subunit, Proc. Natl. Acad. Sci. U.S.A. 1991, 88, 1431-1435.
109. D. L. Boger and D. Yohannes, Total synthesis of deoxybouvardin and RA-VII: macrocyclization via an intramolecular Ullmann reaction, J. Am. Chem. Soc. 1991, 113, 1427-1429.
108. D. L. Boger, Design, synthesis, and evaluation of functional analogs of CC-1065, in Heterocycles in Bio-organic Chemistry, Bergman, J.; Van der Plas, H. C.; Simonyi, M., Eds.; Royal Society of Chemistry: Cambridge, 1991, 103-129.
107. D. L. Boger and S. Nakahara, Diels-Alder reactions of N-sulfonyl 1-aza-1,3-butadienes: development of a synthetic approach to the streptonigrone C ring, J. Org. Chem. 1991, 56, 880-884.
106. D. L. Boger, T. Ishizaki, H. Zarrinmayeh, P. A. Kitos, and O. Suntornwat, A potent, simple derivative of an analog of the CC-1065 alkylation subunit, Bioorg. Med. Chem. Lett. 1991, 1, 55-58.
105. D. L. Boger, T. Ishizaki, H. Zarrinmayeh, S. A. Munk, P. A. Kitos, and O. Suntornwat, Duocarmycin DNA alkylation properties and identification, synthesis and evaluation of agents incorporating the pharmacophore of the duocarmycin alkylation subunit: identification of the CC-1065/duocarmycin common pharmacophore, J. Am. Chem. Soc. 1990, 112, 8961-8971.
104. D. L. Boger and D. Yohannes, Total synthesis of L,L-Isodityrosine and isodityrosine-derived agents: K-13, OF4949-III, and OF4949-IV, J. Org. Chem. 1990, 55, 6000-6017.
103. D. L. Boger, T. Ishizaki, P. A. Kitos, and O. Suntornwat, Synthesis of N-(tert-butyloxycarbonyl)-CBI, CBI, CBI-CDPI1, and CBI-CDPI2: enhanced functional analogs incorporating the 1,2,9,9a-tetrahydrocycloprop[1,2-c]benz[1,2-e]indol-4-one (CBI) left-hand subunit, J. Org. Chem. 1990, 55, 5823-5832.
Correction: J. Org. Chem. 1991, 56, 2946.
102. D. L. Boger, Diels-Alder reactions of azadienes: scope and application, Bull. Soc. Chim., Belg. 1990, 99, 599-615.
101. D. L. Boger and R. J. Mathvink, Tandem free ring expansion and 5-exo-dig 5-hexynyl radical cyclization: a useful approach to fused bicyclic carbocycles, J. Org. Chem. 1990, 55, 5442-5444.
100. D. L. Boger and T. T. Curran, Diels-Alder reactions of 1-aza-1,3-butadienes: room-temperature endo-selective LUMOdiene-controlled [4 + 2] cycloaddition reactions of N-sulfonyl 4-ethoxycarbonyl-1-aza-1,3-butadienes, J. Org. Chem. 1990, 55, 5439-5442.
99. D. L. Boger, T. Ishizaki, H. Zarrinmayeh, P. A. Kitos, and O. Suntornwat, Synthesis and preliminary evaluation of agents incorporating the pharmacophore of the duocarmycin/pyrindamycin alkylation subunit: identification of the CC-1065/duocarmycin common pharmacophore, J. Org. Chem. 1990, 55, 4499-4502.
98. D. L. Boger and C. E. Brotherton-Pleiss, Thermal reactions of cyclopropenone ketals. Key mechanistic features, scope, and application of the cycloaddition reactions of cyclopropenone ketals and -delocalized singlet vinylcarbenes: three-carbon 1,1-/1,3-dipoles; in Advances in Cycloaddition Chemistry, Vol. 2, D. P. Curran, Ed., JAI Press, 1990, 147-219.
97. D. L. Boger, R. J. Wysocki, Jr., and T. Ishizaki, Synthesis of N-phenylsulfonyl-CI, N-(tert-butyloxycarbonyl)-CI, CI-CDPI1, and CI-CDPI2: CC-1065 functional analogs incorporating the parent 1,2,7,7a-tetrahydrocycloprop[1,2-c]indol-4-one (CI) left-hand subunit, J. Am. Chem. Soc. 1990, 112, 5230-5240.
96. D. L. Boger, R. S. Coleman, B. J. Invergo, S. M. Sakya, T. Ishizaki, S. A. Munk, H. Zarrinmayeh, P. A. Kitos, and S. C. Thompson, Synthesis and evaluation of aborted and extended CC-1065 functional analogs: (+)- and (-)-CPI-PDE1; (+)- and (-)-CPI-CDPI1; and (±)-, (+)-, and (-)-CPI-CDPI3. Preparation of key partial structures and definition of an additional functional role of the CC-1065 central and right-hand subunits, J. Am. Chem. Soc. 1990, 112, 4623-4632.
95. D. L. Boger and R. J. Mathvink, Intramolecular acyl radical-alkene addition reactions: macrocyclization reactions, J. Am. Chem. Soc. 1990, 112, 4008-4011.
94. D. L. Boger and R. J. Mathvink, Tandem free radical alkene addition reactions of acyl radicals, J. Am. Chem. Soc. 1990, 112, 4003-4008.
93. D. L. Boger, W. L. Corbett, and J. Mark Wiggins, Room temperature, endo specific 1-aza-1,3-butadiene Diels-Alder reactions: acceleration of the LUMOdiene-controlled [4 + 2] cycloaddition reactions through noncomplementary azadiene substitution, J. Org. Chem. 1990, 55, 2999-3000.
92. D. L. Boger and T. Ishizaki, Resolution of a CBI precursor and incorporation into the synthesis of (+)- and (-)-CBI, (+)- and (-)-CBI-CDPI1, (+)- and (-)-CBI-CDPI2: enhanced functional analogs of (+)-CC-1065. A critical appraisal of the proposed relationship between electrophile reactivity, DNA binding properties, and cytotoxic potency, Tetrahedron Letters 1990, 31, 793-796.
91. D. L. Boger and I. C. Jacobson, Studies on the total synthesis of fredericamycin A. Preparation of key partial structures and development of an intermolecular alkyne-chromium carbene complex benzannulation cyclization approach to the ABCD(E) ring system, J. Org. Chem. 1990, 55, 1919-1928.
90. D. L. Boger and H. Zarrinmayeh, Regiocontrolled nucleophilic addition to selectively-activated p-quinonediimines: alternative preparation of a key intermediate employed in the preparation of the CC-1065 left-hand subunit, J. Org. Chem. 1990, 55, 1379-1390.
89. D. L. Boger, R. S. Coleman, B. J. Invergo, H. Zarrinmayeh, P. A. Kitos, S. C. Thompson, T. Leong, and L. W. McLaughlin, A demonstration of the intrinsic importance of stabilizing hydrophobic binding and noncovalent van der Waals contacts dominant in the noncovalent CC-1065:DNA binding, Chem.-Biol. Interactions 1990, 73, 29-52.
88. D. L. Boger and D. Yohannes, Design and total synthesis of a conformational analog of bouvardin/deoxybouvardin, Synlett 1990, 33-36.
87. D. L. Boger and D. Yohannes, Total synthesis of OF4949-III and OF4949-IV: unusual effects of remote substituents on macrocyclization reaction rates, Tetrahedron Letters 1989, 30, 5061-5064.
86. D. L. Boger, T. Ishizaki, R. J. Wysocki, Jr., S. A. Munk, P. A. Kitos, and O. Suntornwat, Total synthesis and evaluation of (±)-N-(tert-butyloxycarbonyl)-CBI, (±)-CBI-CDPI1, and (±)-CBI-CDPI2: CC-1065 functional analogs incorporating the equivalent 1,2,9,9a-tetrahydrocycloprop[1,2-c]benz[1,2-e]indol-4-one (CBI) left-hand subunit, J. Am. Chem. Soc. 1989, 111, 6461-6463.
85. D. L. Boger and M. Patel, Recent applications of the inverse electron demand Diels-Alder reaction, in Progress in Heterocyclic Chemistry, 1989, Vol. 1, H. Suschitzky and E. F. V. Scriven, Ed., Pergamon Press, 1989, 30-64.
84. D. L. Boger and D. Yohannes, Synthesis of isodityrosine, Tetrahedron Letters 1989, 30, 2053-2056.
83. D. L. Boger and I. C. Jacobson, Studies on the total synthesis of fredericamycin A: development of an intermolecular alkyne-chromium carbene complex cyclization approach to the ABCDE ring system,Tetrahedron Letters 1989, 30, 2037-2040.
82. D. L. Boger and D. Yohannes, Total synthesis of K-13, J. Org. Chem. 1989, 54, 2498-2502.
81. D. L. Boger and R. J. Mathvink, Phenylselenoesters as effective precursors of acyl radicals for use in intermolecular addition reactions, J. Org. Chem. 1989, 54, 1777-1779.
80. D. L. Boger and R. J. Wysocki, Jr., Total synthesis of (±)-N-benzenesulfonyl- and (±)-N-(tert-butyloxycarbonyl)-CI, (±)-CI-CDPI1, and (±)-CI-CDPI2: CC-1065 functional analogs incorporating the parent 1,2,7,7a-tetrahydrocycloprop[1,2-c]indol-4-one (CI) left-hand subunit, J. Org. Chem. 1989, 54, 1238-1240.
79. D. L. Boger and A. M. Kasper, A general solution to implementing the 4p participation of 1-aza-1,3-butadienes in Diels-Alder reactions: inverse electron demand Diels-Alder reactions of a,b-unsaturatedN-phenylsulfonylimines, J. Am. Chem. Soc. 1989, 111, 1517-1519.
78. D. L. Boger and R. J. Wysocki, Jr., A study of the scope of the [4 + 2] cycloaddition reactions of unactivated 1,3-oxazin-6-ones, J. Org. Chem. 1989, 54, 714-718.
77. D. L. Boger and R. S. Coleman, Natural and synthetic antitumor agents: synthetic studies on CC-1065 and functionally-related agents, in Studies in Natural Products Chemistry, Vol. 3, A.-u.-Rahman, Ed., Elsevier Science Publishers, 1989, 301-389.
76. D. L. Boger and K. D. Robarge, A divergent, de novo synthesis of carbohydrates based on an accelerated inverse electron demand Diels-Alder reaction of 1-oxa-1,3-butadienes, J. Org. Chem. 1988, 53, 5793-5796.
75. D. L. Boger, L. R. Cerbone, and D. Yohannes, Oxidative coupling of methyl 6-hydroxyindole-2-carboxylate with primary amines: preparation of 2-substituted methyl pyrrolo[2,3-e]benzoxazole-5-carboxylates, J. Org. Chem. 1988, 53, 5163-5168.
74. D. L. Boger and R. S. Coleman, Total synthesis of (±)-N2-phenylsulfonyl CPI, (±)-CC-1065, (+)-CC-1065, ent-(-)-CC-1065, and the precise, functional agents: (±)-CPI-CDPI2, (+)-CPI-CDPI2, and (-)-CPI-CDPI2, J. Am. Chem. Soc. 1988, 110, 4796-4807.
73. D. L. Boger and Q. Dang, Inverse electron demand Diels-Alder reactions of 2,4,6-tris(ethoxycarbonyl)-1,3,5-triazine and 2,4,6-tris(methylthio)-1,3,5-triazine: pyrimidine introduction, Tetrahedron 1988,44, 3379-3390.
72. D. L. Boger and R. J. Wysocki, Jr., Thermal cycloaddition reactions of p-delocalized singlet vinylcarbenes: three-carbon 1,1-/1,3-dipoles. The thermal three-carbon + two-carbon cycloaddition, J. Org. Chem. 1988, 53, 3408-3421.
71. D. L. Boger and R. J. Mathvink, Acyl radicals: functionalized free radicals for intramolecular cyclization reactions, J. Org. Chem. 1988, 53, 3377-3379.
70. D. L. Boger and K. D. Robarge, Accelerated inverse electron demand Diels-Alder reactions of 1-oxa-1,3-butadienes: [4 + 2] cycloaddition reactions of a,b-unsaturated -keto esters, J. Org. Chem. 1988,53, 3373-3377.
69. D. L. Boger, Diels-Alder reactions of heterocyclic azadienes: development of a strategy for the total synthesis of streptonigrin, lavendamycin, and synthetic quinoline-5,8-quinones, Strategies and Tactics in Organic Synthesis, Vol. 2, T. Lindberg, Ed., Academic Press, 1988, 2-56.
68. D. L. Boger and S. M. Sakya, Inverse electron demand Diels-Alder reactions of 3,6-bis(methylthio)-1,2,4,5-tetrazine: 1,2-diazine introduction and direct implementation of a divergent 1,2,4,5-tetrazine-1,2-diazine-benzene (indoline/indole) Diels-Alder strategy, J. Org. Chem. 1988, 53, 1415-1423.
67. D. L. Boger and M. Patel, Total synthesis of prodigiosin, prodigiosene, and desmethoxyprodigiosin: Diels-Alder reactions of heterocyclic azadienes and development of an effective palladium(II)-promoted 2,2'-bipyrrole coupling procedure, J. Org. Chem. 1988, 53, 1405-1415.
66. D. L. Boger and R. S. Coleman, Total Synthesis of (+)-CC-1065 and ent-(-)-CC-1065, J. Am. Chem. Soc. 1988, 110, 1321-1323.
65. D. L. Boger and R. S. Coleman, Total Synthesis of (+)- and (-)-CPI-CDPI2: (+)-3bR,4aS- and (-)-3bS,4aR-deoxy CC-1065, J. Org. Chem. 1988, 53, 695-698.
64. D. L. Boger and D. Yohannes, Studies on the total synthesis of bouvardin and deoxybouvardin: cyclic hexapeptide cyclization studies and preparation of key partial structures, J. Org. Chem. 1988, 53, 487-499.
63. D. L. Boger, J. S. Panek, and M. Yasuda, Preparation and Diels-Alder reaction of a reactive, electron-deficient heterocyclic azadiene: triethyl 1,2,4-triazine-3,5,6-tricarboxylate, Organic Syntheses 1987,66, 142-150.
62. D. L. Boger and D. Yohannes, Selectively protected L-Dopa derivatives: application of the benzylic hydroperoxide rearrangement, J. Org. Chem. 1987, 52, 5283-5286.
61. D. L. Boger, M. Yasuda, L. A. Mitscher, S. D. Drake, P. A. Kitos, and S. C. Thompson, Streptonigrin and lavendamycin partial structures. Probes for the minimum, potent pharmacophore and chemical mechanism of action of streptonigrin, lavendamycin and synthetic quinoline-5,8-quinones, J. Med. Chem. 1987, 30, 1918-1928.
60. D. L. Boger and M. Yasuda, Streptonigrin and lavendamycin partial structures. Preparation of 7-amino-2-(2'-pyridyl)quinoline-5,8-quinone-6'-carboxylic acid: a probe for the minimum, potent pharmacophore of the naturally occurring antitumor-antibiotics, J. Heterocycl. Chem. 1987, 24, 1253-1260.
59. D. L. Boger and M. Patel, Indole N-carbonyl compounds: preparation and coupling of indole-1-carboxylic acid anhydride, J. Org. Chem. 1987, 52, 3934-3936.
58. D. L. Boger and M. Patel, Total synthesis of prodigiosin, Tetrahedron Letters 1987, 28, 2499-2502.
57. D. L. Boger and M. Patel, Activation and use of pyrrole-1-carboxylic acid in the preparation of pyrrole N-carbonyl compounds: pyrrole-1-carboxylic acid anhydride, J. Org. Chem. 1987, 52, 2319-2323.
56. D. L. Boger and R. S. Coleman, Further observations on the Lewis acid-catalyzed benzylic hydroperoxide rearrangement: use of a boron trifluoride etherate-hydrogen peroxide preformed, aged reagent,Tetrahedron Letters 1987, 28, 1027-1030.
55. D. L. Boger, R. S. Coleman, and B. J. Invergo, Studies on the total synthesis of CC-1065: preparation of a synthetic, simplified 3-carbamoyl 1,2-dihydro[3,2-e]indole dimer/trimer/tetramer (CDPI dimer/trimer/tetramer) and development of methodology for PDE-I dimer methyl ester formation, J. Org. Chem. 1987, 52, 1521-1530.
54. D. L. Boger and R. S. Coleman, Diels-Alder reactions of heterocyclic azadienes: total synthesis of PDE-I, PDE-II, and PDE-I dimer methyl ester, J. Am. Chem. Soc. 1987, 109, 2717-2727.
53. D. L. Boger, C. E. Brotherton, and G. I. Georg, Preparation and three-carbon + two-carbon cycloaddition of a cyclopropenone ketal: cyclopropenone 1,3-propanediyl ketal, Organic Syntheses 1987, 65, 32-40.
52. D. L. Boger and M. D. Mullican, Preparation and inverse electron demand Diels-Alder reaction of an electron-deficient diene: methyl 2-oxo-5,6,7,8-tetrahydro-2H-1-benzopyran-3-carboxylate, Organic Syntheses 1987, 65, 98-107.
51. D. L. Boger and R. S. Coleman, The benzylic hydroperoxide rearrangement: observations on the preparative scope of a convenient alternative to the Baeyer-Villiger rearrangement, J. Org. Chem. 1986,51, 5436-5439.
50. D. L. Boger and R. S. Coleman, Diels-Alder cycloaddition of heterocyclic azadienes: total synthesis of PDE-II methyl ester, J. Org. Chem. 1986, 51, 3250-3252.
49. D. L. Boger, Diels-Alder cycloaddition reactions of heterocyclic azadienes: scope and applications, Chem. Rev. 1986, 86, 781-793.
48. D. L. Boger and C. E. Brotherton, Thermal reactions of cyclopropenone ketals. Application of the cycloaddition reactions of π-delocalized singlet vinylcarbenes: three-carbon 1,1-/1,3-dipoles. An alternative preparation of deacetamidocolchicine: formal total synthesis of colchicine, J. Am. Chem. Soc. 1986, 108, 6713-6719.
47. D. L. Boger and C. E. Brotherton, Thermal reactions of cyclopropenone ketals. Key mechanistic features and scope of the cycloaddition reactions of π-delocalized singlet vinylcarbenes: three-carbon 1,1-/1,3-dipoles, J. Am. Chem. Soc. 1986, 108, 6695-6713.
46. D. L. Boger and C. E. Brotherton, Diels-Alder cycloadditions of cyclopropenone ketals: dual participation in inverse electron demand (LUMOdiene-controlled) and normal (HOMOdiene- controlled) Diels-Alder reactions. Approaches to the preparation of tropones, Tetrahedron 1986, 42, 2777-2785.
45. D. L. Boger and M. Yasuda, An improved and divergent introduction of the streptonigrin and lavendamycin quinoline-5,8-quinone AB ring systems, Heterocycles 1986, 24, 1067-1073.
44. D. L. Boger and M. Patel, Diels-Alder cycloadditions of rigid dienophiles: a probe for allylic axial substituent control of p-facial selectivity in the Diels-Alder reaction, Tetrahedron Letters 1986, 27, 683-686.
43. D. L. Boger, J. S. Panek, S. R. Duff, and M. Yasuda, Total synthesis of lavendamycin methyl ester, J. Org. Chem. 1985, 50, 5790-5795.
42. D. L. Boger, S. R. Duff, J. S. Panek, and M. Yasuda, Inverse electron demand Diels-Alder reactions of heterocyclic azadienes. Studies on the total synthesis of lavendamycin: investigative studies on the preparation of the CDE b-carboline ring system and AB quinoline-5,8-quinone ring system, J. Org. Chem. 1985, 50, 5782-5789.
41. D. L. Boger, J. S. Panek, R. S. Coleman, J. Sauer, and F. X. Huber, A direct, convenient preparation of dimethyl 1,2,4,5-tetrazine-3,6-dicarboxylate, J. Org. Chem. 1985, 50, 5377-5379.
40. D. L. Boger, M. D. Mullican, L. A. Mitscher, S. Drake, and P. A. Kitos, Antimicrobial and antitumor properties of 9,10-dihydrophenanthrenes, structure-activity studies on juncusol, J. Med. Chem. 1985,28, 1543-1547.
39. D. L. Boger and J. S. Panek, Inverse electron demand Diels-Alder reactions of heterocyclic azadienes: formal total synthesis of streptonigrin, J. Am. Chem. Soc. 1985, 107, 5745-5754.
38. D. L. Boger and C. E. Brotherton, Thermal, four-carbon + three-carbon cycloaddition reaction of cyclopropenone ketals. Total synthesis of deacetamidocolchicine: formal total synthesis of colchicine, J. Org. Chem. 1985, 50, 3425-3427.
37. D. L. Boger, M. Patel, and F. Takusagawa, Regioselectivity of the intermolecular Diels-Alder reaction of acyl nitroso compounds (C-nitrosocarbonyl compounds) and nitrosoformates (O-nitrosocarbonyl compounds). Preparation of functionalized, cis-D6-1-octalones, J. Org. Chem. 1985, 50, 1911-1916.
36. D. L. Boger, M. D. Mullican, M. R. Hellberg, and M. Patel, Preparation of optically-active, functionalized cis-Δ6-1-octalones, J. Org. Chem. 1985, 50, 1904-1910.
35. D. L. Boger, C. E. Brotherton, and G. I. Georg, Thermal, three-carbon + two-atom cycloaddition of cyclopropenone ketals with carbon-heteroatom double bonds: preparation of butenolides, furans, and γ-keto esters, Tetrahedron Letters 1984, 25, 5615-5619.
34. D. L. Boger and C. E. Brotherton, Effective, thermal one-carbon + two-atom cycloaddition of cyclopropenone ketals with electron-deficient olefins: cyclopropane formation, Tetrahedron Letters 1984, 25, 5611-5614.
33. D. L. Boger, R. S. Coleman, J. S. Panek and D. Yohannes, Thermal cycloaddition of dimethyl 1,2,4,5-tetrazine-3,6-dicarboxylate with electron-rich olefins: 1,2-diazine, pyrrole introduction. Preparation of octamethylporphin (OMP), J. Org. Chem. 1984, 49, 4405-4409.
32. D. L. Boger and M. Patel, Regioselectivity of the intermolecular Diels-Alder reaction of acyl nitroso compounds (C-nitrosocarbonyl compounds) and nitrosoformates (O-nitrosocarbonyl compounds), J. Org. Chem. 1984, 49, 4098-4099.
31. D. L. Boger and J. S. Panek, Palladium(0)-mediated β-carboline synthesis: preparation of the CDE ring system of lavendamycin, Tetrahedron Letters 1984, 25, 3175-3178.
30. D. L. Boger, C. E. Brotherton, J. S. Panek, and D. Yohannes, Direct introduction of nitriles via use of unstable Reissert intermediates: convenient procedures for the preparation of 2-cyanoquinolines and 1-cyanoisoquinolines, J. Org. Chem. 1984, 49, 4056-4058.
29. D. L. Boger and C. E. Brotherton, Total synthesis of azafluoranthene alkaloids: rufescine and imelutine, J. Org. Chem. 1984, 49, 4050-4055.
28. D. L. Boger and M. D. Mullican, Regiospecific total synthesis of juncusol, J. Org. Chem. 1984, 49, 4045-4049.
27. D. L. Boger and M. D. Mullican, Inverse electron demand Diels-Alder reactions of 3-carbomethoxy-2-pyrones. Controlled introduction of oxygenated aromatics: benzene, phenol, catechol, resorcinol, pyrogallol annulation. Regiospecific, total synthesis of sendaverine and a preparation of 6,7-benzomorphans, J. Org. Chem. 1984, 49, 4033-4044.
26. D. L. Boger and R. S. Coleman, Intramolecular Diels-Alder reactions of 1,2-diazines: general indoline synthesis. Studies on the preparation of the central and right-hand segment of CC-1065, J. Org. Chem. 1984, 49, 2240-2245.
25. D. L. Boger and C. E. Brotherton, Effective, thermal three-carbon + two-carbon cycloaddition for cyclopentenone formation: formal 1,3-dipolar cycloaddition of cyclopropenone ketals, J. Am. Chem. Soc.1984, 106, 805-807.
24. D. L. Boger and M. D. Mullican, Inverse electron demand Diels-Alder reactions of 3-carbomethoxy-2-pyrones. Controlled introduction of oxygenated aromatics: benzene, phenol, catechol, resorcinol, and pyrogallol annulation, Tetrahedron Letters 1983, 24, 4939-4942.
23. D. L. Boger and J. S. Panek, 1,2,4-Triazine preparation via thermal cycloaddition of 3,6-dicarbomethoxy-1,2,4,5-tetrazine with aryl thioimidates, Tetrahedron Letters 1983, 24, 4511-4514.
22. D. L. Boger, Diels-Alder reactions of azadienes, Tetrahedron 1983, 39, 2869-2939. Tetrahedron Report No. 153.
21. D. L. Boger and J. S. Panek, Formal total synthesis of streptonigrin, J. Org. Chem. 1983, 48, 621-623.
20. D. L. Boger and J. S. Panek, Pyridine construction via thermal cycloaddition of 1,2,4-triazines with enamines: studies on the preparation of the biaryl CD ring system of streptonigrin, J. Org. Chem. 1982,47, 3763-3765.
19. D. L. Boger, M. D. Mullican, and M. Patel, Synthetic analgesics: preparation of 6,7-benzomorphans, Tetrahedron Letters 1982, 23, 4559-4562.
18. D. L. Boger and M. D. Mullican, Regiospecific total synthesis of juncusol, Tetrahedron Letters 1982, 23, 4555-4558.
17. D. L. Boger and M. D. Mullican, Inverse electron demand Diels-Alder reaction of 3-carbomethoxy-2-pyrones with 1,1-dimethoxyethylene: a mild method of aryl introduction, Tetrahedron Letters 1982,23, 4551-4554.
16. D. L. Boger, J. Schumacher, J. S. Panek, M. D. Mullican, and M. Patel, Thermal cycloaddition reaction of 1,3,5-triazine with enamines: regiospecific pyrimidine annulation, J. Org. Chem. 1982, 47, 2673-2675.
15. D. L. Boger, J. S. Panek, and M. M. Meier, Diels-Alder reaction of heterocyclic azadienes, 2. Catalytic Diels-Alder reactions of in situ generated enamines with 1,2,4-triazines: general pyridine annulation,J. Org. Chem. 1982, 47, 895-897.
14. D. L. Boger, C. E. Brotherton, and M. D. Kelley, A simplified isoquinoline synthesis, Tetrahedron 1981, 37, 3977-3980.
13. D. L. Boger and C. E. Brotherton, Preparation of 3-vinyl-4-methyl-1,2,5-oxadiazole and 3-vinyl-4-methyl-1,2,5-thiadiazole, J. Heterocyclic Chem. 1981, 18, 1247-1249.
12. D. L. Boger and J. S. Panek, Diels-Alder reaction of heterocyclic azadienes, I. Thermal cycloaddition of 1,2,4-triazine with enamines: a simple preparation of substituted pyridines, J. Org. Chem. 1981, 46, 2179-2182.
11. D. L. Boger and J. S. Panek, Preparation of (R)- and (S)-4-iodo-1,2-epoxybutane, (R)- and (S)-(2-iodoethyl)oxirane, useful chiral synthons, J. Org. Chem. 1981, 46, 1208-1210.
10. D. L. Boger and M. D. Mullican, A simple and convenient phenol annulation, J. Org. Chem. 1980, 45, 5002-5004.
9. D. L. Boger, Part I: New annulation processes, Part II: Studies directed toward a biomimetic synthetic approach to prostaglandins, Ph.D. dissertation, Harvard University, February 1980, 238 pp.
8. D. L. Boger, A convenient preparation of 2-substituted benzothiazoles, J. Org. Chem. 1978, 43, 2296-2297.
7. E. J. Corey and D. L. Boger, A new and unusually flexible scheme for annulation to form fused cyclohexanone units, Tetrahedron Letters 1978, 4597-4600.
6. E. J. Corey and D. L. Boger, Oxidative cationic cyclization reactions effected by pyridinium chlorochromate, Tetrahedron Letters 1978, 2461-2464.
5. D. L. Boger, Concerning the stereochemistry of 9-methyl-D1-3-octalone, Tetrahedron Letters 1978, 17-18.
4. E. J. Corey and D. L. Boger, New annulation processes for fused and spiro rings based on the chemistry of benzothiazoles, Tetrahedron Letters 1978, 13-16.
3. E. J. Corey and D. L. Boger, Application of benzothiazoles to carbon-carbon bond formation in organic synthesis, Tetrahedron Letters 1978, 9-13.
2. E. J. Corey and D. L. Boger, Benzothiazoles as carbonyl equivalents, Tetrahedron Letters 1978, 5-8.
1. A. W. Burgstahler, D. L. Boger and N. K. Naik, Synthesis and chiroptical properties of some asymmetric planar cisoid dienes, Tetrahedron 1976, 32, 309-315.
Books
Hetero Diels-Alder Methodology in Organic Synthesis, D. L. Boger and S. M. Weinreb, Academic Press: San Diego, 1987.
Modern Organic Synthesis: Lecture Notes, Dale L. Boger, TSRI Press: San Diego, 1999.



























