|
(page 2 of 2)
Shutting Down the Cancer Genes erbB-2 and erbB-3
In a series of reports last year, Barbas demonstrated the
efficacy of using multiple zinc finger proteins to bind to
two 18 base pair sequences in the promotor regions of the
protooncogenes erbB-2 and erbB-3.
These two genes are involved in human cancers, particularly
breast and ovarian cancers, and show increased expression
in cancerous cells.
By fusing a set of zinc fingers that bound to the regulatory
regions of these genes with the repressor protein KRAB, they
were able to shut off expression of erbB-2 and erbB-3. Then,
by fusing the transactivator protein VP64, they were able
to dramatically increase the transcription of the genes.
"We’re also using the approach to target sickle-cell
anemia," says Barbas.
Sickle-cell anemia is a chronic disease prevalent in individuals
of African, Mediterranean, or Southwest Asian origin where
the oxygen binding protein hemoglobin inside red blood cells
have a single amino acid mutation that causes them to polymerize,
which in turn causes the red blood cells to become misshapen
into "sickles." This condition can lead to clotting, vascular
damage, thrombosis, and extensive tissue and bone damage in
adults.
All humans have a redundant hemoglobin gene, however. The
fetal hemoglobin gene is always normal in adults with the
sickle cell mutation, and as a result, people do not suffer
from sickle-cell anemia as infants. At some point in early
development, though, the responsibility for producing the
body’s hemoglobin switches from the fetal gene to the
adult gene. A number of drugs already exist that increase
the ratio of fetal to adult hemoglobin in the body, but they
are all rather toxic.
"If we can make a genetic switch that turns on fetal hemoglobin,"
says Barbas, "then we can deliver it the same way we can deliver
other genes [into stem cells]." Because zinc finger transcription
factors are rather small proteins, several of them can be
inserted at once, and another switch could be delivered to
simultaneously turn off the bad or defective adult hemoglobin
gene.
This sort of therapy would seek to insert the gene for the
zinc finger regulatory protein into stem cells of a patient
suffering from sickle-cell anemia. Then these stem cells would
grow into red blood cells that would express the normal, fetal
hemoglobin.
The advantage of a stem cell-based therapy is that it could
be done once and the gene correction would be in place forever.
A Potential Strategy Against HIV
This sort of therapy could prove useful as a way of treating
people who are infected with HIV as well. The Barbas laboratory
has collaborated with Associate Professor Bruce Torbett on
an effort to develop a method for delivering therapeutic genes
into patients' stem cells. See
A Vaccine Factory Inside Each Cell.
In that effort, Barbas and his group isolated an antibody,
and its gene, that is specific for CCR5—a coreceptor
that is essential for viral entry. They then designed a peptide
"anchor" on one end that retains it in the cell’s endoplasmic
reticulum. These intracellular antibodies have been termed
"intrabodies." Once the intrabody gene is incorporated in
a cell, the cell would express the intrabody that would then
grab the CCR5 and keep it from getting to the surface of the
cell, effectively preventing HIV from entering the cell.
But HIV has no proofreading mechanism to correct mistakes
when it transcribes itself, and this makes the virus notoriously
error prone, encoding one mutation with every copy. This error
rate is one of the confounding problems of HIV therapy, of
course, because mutations in the gene products that are themselves
targets of the therapy can reduce the affinity—and therefore
the efficacy—of the drug. For instance, HIV can stop
using the CCR5 coreceptor and switch to the CXCR4 coreceptor,
which cannot be knocked out.
However, mutations that knock out the expression of essential
genes are lethal to the virus. Fortunately, certain parts
of the viral genome do not tolerate mutations—they are
completely conserved.
Barbas now believes the same stem cells can also be modified
so that if the virus does mutate and does get in, it can still
be blocked through the action of zinc finger transcription
factors designed to target these conserved regions.
For instance, the cells could be equipped with proteins
that, when expressed, could bind to the conserved long terminal
repeat regions of the HIV genome, which are the ends that
integrate into the host cell's DNA. Zinc fingers bound at
these sites should prevent the DNA from inserting.
"We're looking towards building those fingers now," says
Barbas.
Another conserved region is the primer binding site, which
is the 21 base pair site at the front of the HIV genome where
reverse transcription of the viral RNA into viral DNA starts.
After a virion enters a cell and uncoats, the RNA must attach
to a human tRNA in order to turn on the reverse transcription.
In fact this is the most conserved region in the HIV genome.
"Since the human tRNA is not changing, the virus doesn't change,"
says Barbas. "If the primer binding site changes, then the
virus cannot replicate in human cells."
Targeting this site with a zinc finger fused to a repressor
protein should stop viral replication.
"Ideally, we will be able to make multiple blockades of
the HIV virus," Barbas says. The CCR5 blockers would prevent
the HIV from entering cells. The LTR binding fingers, if Barbas
is successful in designing them, could prevent the virus from
integrating into the host cell genome, and the primer binding
site repressors could shut down replication.
"So if we don't get it coming in, at the surface, or before
it gets into the nucleus," says Barbas, "we’ll inhibit
stop it once it tries to make viral RNA."
1 | 2 |

|
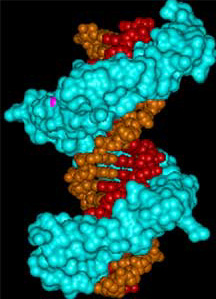
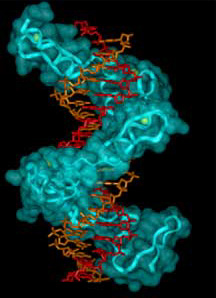
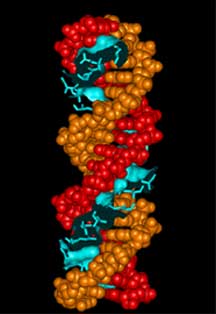
Three views of zinc fingers bound to
DNA. Top panel shows the space filling model of the zinc fingers
(in blue) bound to the double stranded DNA (in red and orange).
Middle panel shows space filling model with backbone ribbon.
Bottom panel shows a partial space filling model of areas
of contact with side chains shown as worms.
For more information, visit:
|

