New Study Shines Light on Mutations Responsible for Debilitating Heart Conditions
The new insights could aid in the development of drug therapies to strengthen the hearts of patients suffering from age-related heart failure.
JUPITER, FL – August 11, 2016 – The leading cause of death in the world remains cardiovascular diseases, which are responsible for more than one third of overall mortality, according to the World Health Organization. Obesity and diet are obvious culprits behind heart disease but, over the past decade, research has also pointed to genetic factors, specifically mutations in cell adhesion components—the forces that bind cells together.
In a new study, scientists from the Florida campus of The Scripps Research Institute offer new molecular insights into how the interaction between specific genetic mutations and a cytoskeletal protein critical for the proper development and maintenance of heart tissue can lead to conditions such as dilated cardiomyopathy (DCM) and hypertrophic cardiomyopathy (HCM)—and ultimately heart failure.
The new study, which was led by Associate Professor T. Izard of the Florida campus of TSRI, is published this week in an early online edition of the journal Proceedings of the National Academy of Sciences. The new insights could aid in the development of drug therapies to strengthen the hearts of patients suffering from age-related heart failure.
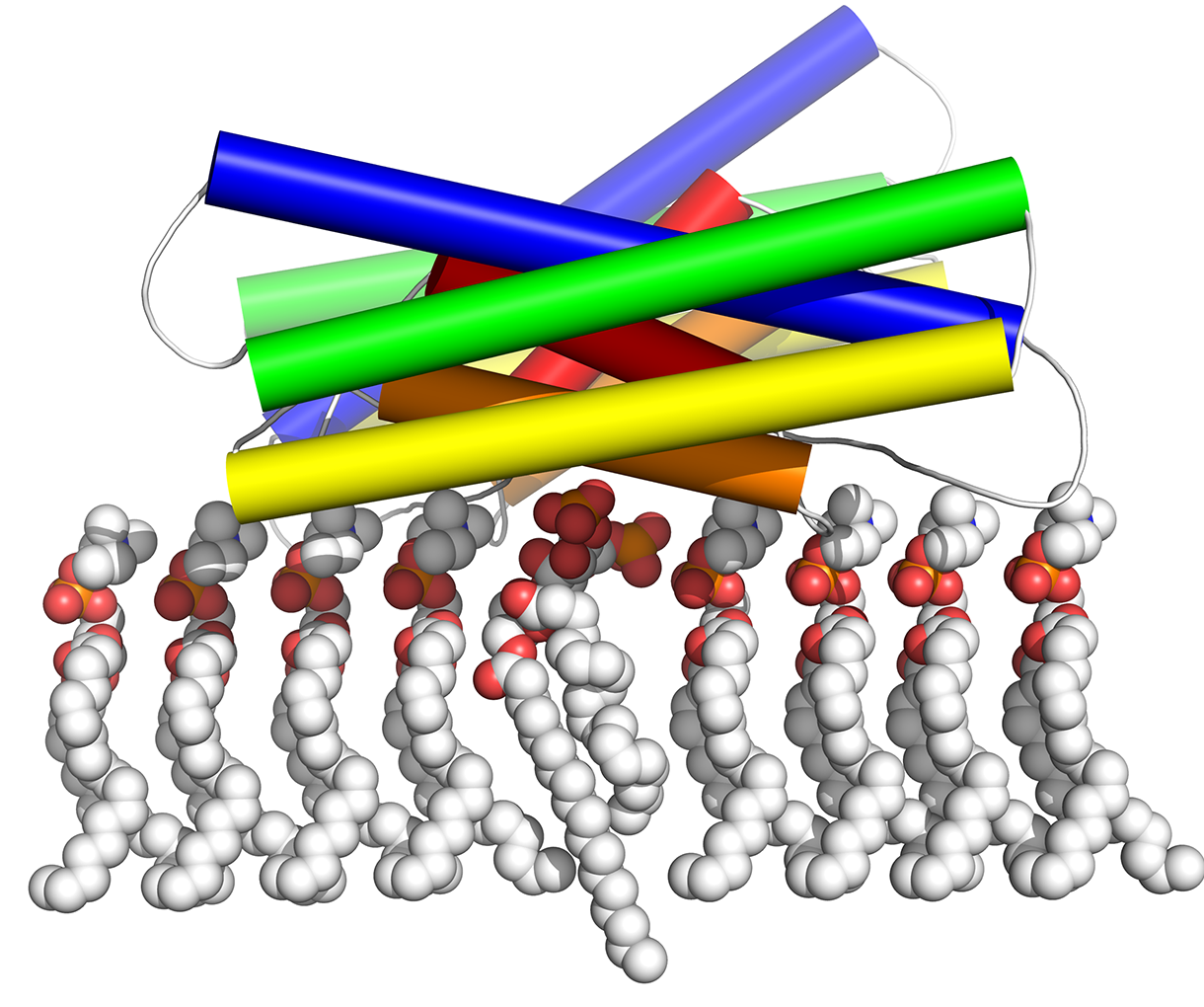
The study focuses on the protein vinculin and a variant form known as metavinculin, which is found only in muscle tissue. Vinculin has been shown to reinforce the myocardial cell cytoskeleton, improving heart muscle contractility and prolonging life, while metavinculin plays an essential role in the development and function of the heart.
Both vinculin and metavinculin regulate cell adhesion and migration by linking the cell’s cytoskeleton to adhesion receptor complexes via a process known as dimerization—the joining of two similar subunits. Control of the dimerization process is crucial for normal protein function in cell adhesion sites.
But mutations in the variant metavinculin, either inherited or spontaneous, corrupt this process, altering dimerization and, the study suggests, producing a decreased ability to stabilize critical cell adhesions, weakening the heart muscle over time.
The researchers found that these mutations—specifically, a mutation known as R975W in metavinculin—dictate the type of interaction during dimerization and can actually block the process. That, in turn, results in heart muscles that are far more susceptible to stress-induced heart disease.
The first author of the study, “Differential Lipid Binding of Vinculin Isoforms Promotes Quasiequivalent Dimerization,” is Krishna Chinthalapudi of TSRI. Other authors include Erumbi S. Rangarajan of TSRI and David T. Brown of the University of Mississippi Medical Center.
The laboratory is supported by the National Institutes of Health, the Department of Defense, the American Heart Association and the State of Florida.
For more information, contact press@scripps.edu See More News

