|
Stem and Endothelial Cell Biology
Nothing more dramatically captures the imagination of the visually
impaired patient or the ophthalmologist treating them than the possibility of rebuilding a damaged retina with "stem cells." Defined as pluripotent cells capable of differentiating into a variety of cell types, stem cells can be derived from early embryos and, under appropriate conditions, will differentiate into a variety of tissues including muscle, kidney, brain, blood, liver, skin and retina. Stem cells have also been identified and isolated from adult tissues and presumably represent a pool of progenitor cells that may serve to maintain a supply of cells to maintain various tissue types as well as rescue/repair damaged tissue following injury or stress.
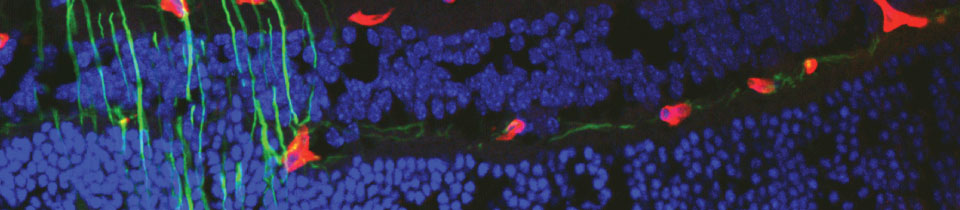
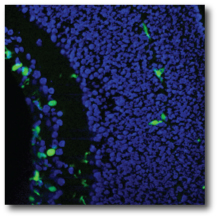
Adult bone marrow derived stem cells. Adult bone marrow is known to contain a population of hematopoietic stem cells (HSCs) that can be divided into lineage positive (Lin+) and lineage negative (Lin-) subpopulations with regard to their potential to differentiate into formed elements of the blood. The Lin- population contains a variety of progenitor cells including those capable of becoming vascular endothelial cells. These endothelial progenitor cells mobilize from the bone marrow in response to a variety of signaling molecules and can target sites of angiogenesis in ischemic peripheral vasculature, myocardium, or induced ocular injury. We have reported that Lin- HSCs injected directly into the eye can target activated astrocytes, a hallmark of many ocular vascular and degenerative diseases, and participate in normal developmental angiogenesis in neonatal mice or injury-induced neovascularization in the adult. We have also reported that intravitreally injected adult bone marrow-derived Lin- HSCs, can completely prevent retinal vascular degeneration ordinarily observed in the rd1 or rd10 mouse models of retinal degeneration, and this vascular rescue correlates with neuronal rescue. Large scale genomic analysis of rescued eyes revealed significant upregulation of anti-apoptotic genes and this effect is also observed when human bone marrow-derived Lin- HSCs are used to treat severe combined immunodeficient (SCID) mice with retinal degeneration. In addition, these HSCs, if previously transfected with a plasmid encoding a secreted anti-angiogenic peptide, will profoundly inhibit retinal angiogenesis suggesting that cell-based therapy may be useful in the inhibition of proliferative retinopathies.
The specific identity of these pluripotent cells remain an enigma. HSC can differentiate into a variety of cell types other than hematopoietic cells, including neurons, glia, and muscle, depending on their environment. The observation that HSCs contain a pool of EPC capable of incorporating into retinal vasculature has recently been demonstrated. Similarly, systemically administered HSC can function as hemangioblasts during hypoxia-stimulated retinal neovascularization as well as contribute to laser-stimulated choroidal neovascularization in a mouse model. A recent study from our laboratory demonstrates a role of R-cadherin in the targeting of HSC to the retinal vasculature. Thus, inhibition of targeting of circulating HSC that may contribute to abnormal ocular neovascularization may, in fact, reduce the incidence of such new blood vessel growth. Unfortunately, inhibition of neovascularization under such ischemic conditions may serve to promote ongoing ischemia; would it be better to coax the newly forming vessels into functional ones that could alleviate hypoxia? Our study suggests that this, in fact, may be possible. The HSC fraction used in these experiments not only inhibited angiogenesis when engineered to express an anti-angiogenic, but also rescued and stabilized (e.g., "matured") degenerating vessels. More surprisingly, we observed that by preventing vascular degeneration there was also a trophic effect on the photoreceptors themselves, suggesting that autologous bone marrow grafts of HSC fractions containing EPC may provide trophic effects on associated neural tissue that goes beyond simple nutrition. Such observations could provide a rationale for the use of HSC in the treatment of a variety of inherited retinal degenerations such as retinitis pigmentosa.
The potential use of HSC for cell based therapies in the eye presents many challenges. Transfection efficiencies are improving with newer Lenti- and Adenoviral, as well as non-viral, vectors. Another enigma in the stem cell field is the issue of HSC "homing"; all of the molecular signals involved have not yet been identified, but a better understanding of these signals would be of immense benefit in exploiting the potential use of HSC in therapeutic angiogenesis as well as directed cell therapy. Finally, which cell type in the Lin-HSC actually adheres to astrocytes and incorporates into, and/or selectively associates with, the developing vasculature? Many of the surface markers routinely used to identify cell populations vary from laboratory to laboratory and, certainly, expression of these markers depends on the environment into which the cells are placed. Thus, sorting cells based on cell surface antigen expression too early in their lineage may lead to cases of mistaken identity or loss of potentially useful cells. Functional definition of stem cell populations, as we have reported, provides an assessable endpoint but warrants further investigation. Finally, the use of stem cells as therapeutic vehicles has the potential to selectively and potently deliver drugs to the back of the eye in physiologically meaningful doses. Since this is where most vision-threatening disease occurs, the use of these cells holds great promise for millions of patients suffering from currently untreatable conditions.
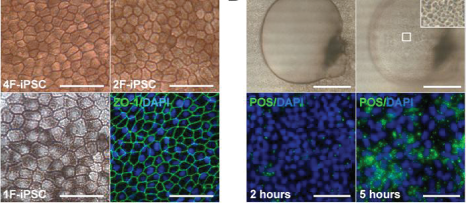
Induced pluripotent stem cells (iPSC) as a source of autologous RPE grafts. Generating RPE from hESC to serve as healthy grafts to replace diseased RPE in patients with AMD has merit, but there are limitations in this approach. There is considerable variability in the quality of the RPE obtained from different hESC lines and using fresh fetal material has obvious limitations. More importantly, the RPE obtained from hESC would not be autologous and require growth on mouse feeder cell layers and/or matrigel, conditions shown to be associated with retained murine antigenicity. Subretinal implantation of such cells has led to rapid and pronounced graft rejection. One way to circumvent long-term systemic immunosuppression following RPE transplantation is to use autologous somatic cells induced to form pluripotent stem cells (iPSC) from which the RPE could be derived. Recent advances in stem cell biology have demonstrated that iPSC can be derived from somatic cells such as skin fibroblasts by using retroviral vectors to introduce reprogramming factors (Oct4, Sox2, Nanog, Lin28) into somatic cells. However, random viral integration and spontaneous reactivation of reprogramming factors during differentiation pose a cancer risk and limits the potential use of iPSC derived in this manner in human therapy. In addition to this risk, therapeutic application of current technology is also limited by its low efficiency and slow kinetics. Finally, there is the requirement for numerous growth factors and animal cell feeder layers to obtain even limited numbers of iPSC. To overcome these hurdles, increase the efficiency of generating iPSC-derived RPE, and to avoid using lentiviral vectors, we have used small molecules from a unique chemical library and/or an episomal expression vector that simultaneously expresses the four human factors necessary to induce iPSC from somatic cells. We have evaluated the differentiation capacity of iPSCs generated by lentiviral transduction with only two factors or one factor (OCT4/KLF4 or OCT4 only, respectively) and small molecules. Human iPSCs reprogrammed by conventional methods and primary human RPE were used for comparison. Four, two, and one factor-derived iPS (4F-, 2F-, and 1F-iPSCs, respectively) could be differentiated into pigmented cells expressing RPE-specific markers (e.g., bestrophin, CRALBP, RPE65, tyrosinase), exhibiting characteristic RPE morphology, forming polarized monolayers, transporting fluid vectorially, and phagocytosing photoreceptor outer segments. Importantly, 1F-iPS-RPE strongly resembles primary human fetal RPE based on untargeted metabolomic and antibody-based proteomic analyses. Additionally, novel in vivo imaging techniques were employed to demonstrate that 1F-iPS-RPE retard retinal degeneration after subretinal transplantation in the Royal College of Surgeons (RCS) rat, an animal model of RPE-mediated retinal degeneration. Compared to conventional iPSCs, RPE grafts derived from 1F-iPS-RPE may be superior for clinical use due to their reduced number of potentially oncogenic reprogramming factors.