Ongoing Collaboration Between TSRI and Princeton Scientists
Reveals Details of Natural Killer T Cells
By Jason Socrates
Bardi
In the latest of what has become a series of collaborative
studies between two laboratories on opposite coasts, Associate
Professor Luc Teyton of The Scripps Research Institute (TSRI)
and Albert Bendelac of Princeton University further characterize
an immune cell, known as a natural killer (NK) T cell, or
"NK1.1 T cells," which regulate other immune cells.
In the current study, the researchers provide a detailed
look at the maturation of the NK1.1 T cell lineage, following
its migration from its early precursor days in the thymus
to its differentiation in various tissues in the body.
"We are basically trying to understand how the NK1.1 system
works," says Teyton, who adds that the long-standing collaboration
has its roots in Paris, where Teyton and Bendelac knew each
other as students before both came to the United States.
The NK1.1 T cells are unusual in that they fall somewhere
between innate and adaptive immunity. These cells arise in
the thymus. As mature cells, they stimulate an adaptive immune
response and regulate a range of disease states, including
diabetes, cancer, and pathogenic infections.
Like other T cells, they express T cell receptors (TCR)—although
without the normal antigenic variability. However, NK1.1 T
cells also express the "NK" innate immune cell receptors and
may have the ability to see some of the lipids that many bacteria
display on their outer surface. This NK receptor recognizes
the CD1 receptor, which is associated with the major histocompatability
complex on antigen presenting cells, which are like the buglers
that warn the immune system that a pathogen is invading.
NK1.1 T cells bind to a cell surface protein called CD1
that bears an unknown lipidic ligand, one focus of Teyton
and Bendelac's current investigations.
Once the NK1.1 T cells bind to CD1, they become activated
and begin to secrete a large amount of the protein interleukin-4,
which in turn activates helper T cells. The helper T cells
then induce specific B cells to unload bursts of soluble antibodies
into the bloodstream, and these antibodies ultimately deal
with cancerous cells and pathogens.
One of the toughest challenges in studying NK1.1 T cells
is simply expressing and purifying biologically active CD1
receptors. This task is almost impossible in bacterial expression
systems. So Teyton expresses the molecules in Drosophilla
cells and purifies them as empty, fully folded proteins from
culture supernatants. They are subsequently multimerized and
loaded with antigenic lipids to make them react with the T
cell receptor of NK1.1 T cells.
In the current study, Teyton made these reagents to look
specifically at the thymic development of these cells.
"We also expressed NK1.1 soluble T cell receptors to examine
their binding to CD1 molecule," says Teyton. He adds that
these studies are the preliminary work that will lead to the
determination of TCR/CD1 complex structures. The same approach
was used in 1996 by Teyton and TSRI Professor Ian Wilson of
the Department of Molecular Biology, when they crystallized
the TCR and TCR/MHC complex and solved their structures.
The article, "A Thymic Precursor to the NKT Cell Lineage,"
is authored by Kamel Benlagha, Tim Kyin, Andrew Beavis, Luc
Teyton, and Albert Bendelac and appears in the March 14, 2002
issue of the journal Science

|
mCD1d-aGC tetramer
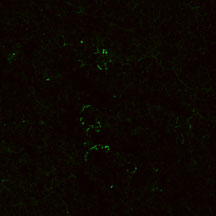
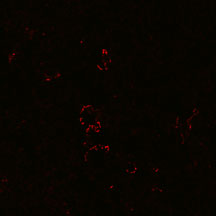
NK1.1 mAb
Merge
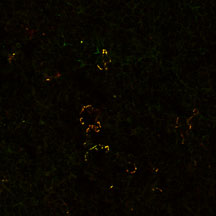
Identification of NK1.1 cells in 7 wk
C57BL/6 mouse lymph nodes by immunocytochemistry. Staining
with CD1-aGC
tetramers (top) superimposes with anti-NK1.1 staining (center).
Observation was carried out by Confocal microscopy (63x (oil)
lens, 1x Zoom).
|

