

Tweet
AIDS Researchers Isolate New Potent and Broadly Effective Antibodies Against HIV
A team of researchers at and associated with the International AIDS Vaccine Initiative (IAVI), The Scripps Research Institute, the biotechnology company Theraclone Sciences, and Monogram Biosciences Inc., a LabCorp company, report in the current issue of Nature the isolation of 17 novel antibodies capable of neutralizing a broad spectrum of variants of HIV, the virus that causes AIDS.
The new antibodies, large protein molecules that bind to pathogens and flag them for destruction, were isolated from blood serum samples collected in a continuing global search for broadly neutralizing antibodies launched by IAVI. They should provide researchers with a new set of targets for the design of vaccine candidates that can elicit similar antibodies to protect people from contracting HIV. Some of the broadly neutralizing antibodies blocked HIV infection of cells as much as 10 to 100 times as potently as previously discovered broadly neutralizing antibodies.
"Most antiviral vaccines depend on stimulating the antibody response to work effectively," said Dennis Burton, a professor at The Scripps Research Institute in La Jolla, CA, and director of the IAVI Neutralizing Antibody Center there. Professor Burton, one of the senior authors of the study, is also a member of the Ragon Institute, in Cambridge, MA. "Because of HIV's remarkable variability, an effective HIV vaccine will probably have to elicit broadly neutralizing antibodies. This is why we expect that these new antibodies will prove to be valuable assets to the field of AIDS vaccine research."
Only a minority of people who are HIV-positive begin to produce broadly neutralizing antibodies after several years of infection. Animal studies suggest that such antibodies could block HIV infection if they were elicited by a preventive vaccine. Researchers prize broadly neutralizing antibodies because their structural and biochemical analysis can reveal how to achieve a preventive vaccine. Specifically, scientists expect that they can use information about how broadly neutralizing antibodies bind to HIV to construct immunogens—the active ingredients of vaccines—that elicit similar antibodies. The potency of broadly neutralizing antibodies matter because a highly potent antibody could confer such protection at relatively low levels.
"Solving the neutralizing antibody problem is perhaps the greatest challenge facing the field today," said IAVI's chief scientific officer, Wayne Koff. "IAVI concluded many years ago that unlocking the information stored in broadly neutralizing antibodies was going to be essential to the fulfillment of our mission—ensuring the design and development of broadly effective AIDS vaccines. This is why we support several laboratories around the world that are designing novel vaccine candidates on the basis of what we're learning from such antibodies. We have no doubt that these new broadly neutralizing antibodies will contribute a great deal to our own immunogen design efforts and, we hope, those of other researchers working on AIDS vaccines."
In that regard, the new broadly neutralizing antibodies are encouraging. Many of them bind hitherto unknown molecular structures, or epitopes, on the surface of HIV. This means that they could significantly broaden the target options researchers have in designing vaccines to elicit similar antibodies.
How the Antibodies Were Discovered
The 17 new broadly neutralizing antibodies described in the current Nature report were isolated from four HIV-positive individuals. The effort, sponsored by IAVI, is unprecedented in scale and distinguished by its emphasis on identifying antibodies that neutralize subtypes of HIV circulating primarily in developing countries. It had previously yielded three potent broadly neutralizing antibodies, two of which, PG9 and PG16, were isolated by this research team in 2009 and described in the journal Science. Another broadly neutralizing antibody was subsequently isolated from these samples by researchers at the Vaccine Research Center of the National Institutes of Health, who have also discovered a set of broadly neutralizing antibodies from separate blood samples using an entirely different approach.
Both the previous and current studies used Theraclone Science's highly sensitive I-STAR™ technology to isolate the antibodies. The new crop of broadly neutralizing antibodies, like PG9 and PG16, was rescued from cell cultures derived from single antibody-producing B cells used for antibody discovery and development. Theraclone Sciences Executive Chair and Interim CEO Steven Gillis commented, "We're delighted that I-STAR has provided essential support in identifying broadly neutralizing antibodies that will contribute to advancing AIDS vaccine development. In this project, and in our own infectious disease and cancer programs, the I-STAR platform continues to demonstrate a remarkably powerful ability to isolate rare antibodies with unique properties. Theraclone values these collaborative opportunities in which I-STAR can be used to help improve treatment for critical diseases."
Monogram Biosciences, which also participated in the discovery of PG9 and PG16, conducted the neutralization assays essential to isolating the new broadly neutralizing antibodies. The serum samples from which they were isolated represent the top one percent of all such samples gathered by IAVI and its partners, in terms of the number of HIV variants they neutralize and the potency with which they do so.
"Monogram has developed a highly skilled scientific team capable of taking on a variety of biomedical challenges," said Chris Petropoulos, vice president, Laboratory Corporation of America Holdings, Research and Development, Monogram Biosciences. "Their expertise and innovation has been invaluable to the discovery of these new antibodies. This research illustrates the important role different sectors of the research and health care community can play in supporting global health initiatives."
The analysis of the new antibodies also hints at how future vaccines ought to be formulated to maximize their effectiveness. On the basis of their analyses, the authors of the report conclude that AIDS vaccine candidates that seek to effectively harness the antibody response should probably attempt to elicit certain combinations of broadly neutralizing antibodies if they are to provide truly comprehensive protection from HIV.
The first authors of the study, "Broad neutralization coverage of HIV by multiple highly potent antibodies," are Laura M. Walker of Scripps Research and IAVI, and Michael Huber and Katie J. Doores of Scripps Research and Ragon Institute. In addition to Burton, Koff, Walker, Huber, Doores, and protocol investigators listed in the supporting online material, authors of the study include: Emilia Falkowska of Scripps Research and Ragon Institute; Robert Pejchal and Pascal Poignard of Scripps Research and IAVI; Jean-Philippe Julien, Sheng-Kai Wang, Alejandra Ramos, Chi-Huey Wong, and Ian A. Wilson of Scripps Research; Po-Ying Chan-Hui, Matthew Moyle, Jennifer L. Mitcham, Phillip W. Hammond, and Ole A. Olsen of Theraclone Sciences; Pham Phung and Terri Wrin of Monogram Biosciences; Steven Fling, Sanjay Phogat, and Melissa D. Simek. For more information, see http://www.nature.com/nature/journal/vaop/ncurrent/abs/nature10373.html.
This work was supported by the International AIDS Vaccine Initiative, the National Institute of Allergy and Infectious Diseases of the National Institutes of Health, the Ragon Institute, and the United States for International Development.
Send comments to: mikaono[at]scripps.edu
 "We expect that these new antibodies will prove to be valuable assets to the field of AIDS vaccine research," said Scripps Research Professor Dennis Burton, who is director of the IAVI Neutralizing Antibody Center. (Photo by Kevin Fung.)
"We expect that these new antibodies will prove to be valuable assets to the field of AIDS vaccine research," said Scripps Research Professor Dennis Burton, who is director of the IAVI Neutralizing Antibody Center. (Photo by Kevin Fung.)

The first authors of the new study are Katie Doores (back), Laura Walker (front), and Michael Huber (not shown). (Photo by Cindy Brauer.)
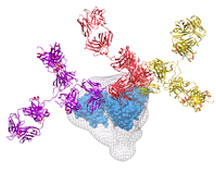
The discovery of new broadly neutralizing antibodies possessing unprecedented potency reveals hitherto unknown molecular structures, or epitopes, on the surface of HIV. This could significantly broaden the target options researchers have in designing vaccines. (Image by Jean-Philippe Julien and Christina Corbaci.)
