

Team Develops Technique to Determine Ethnic Origin of Stem Cell Lines
By Mika Ono
An international team of scientists led by researchers at The Scripps Research Institute has developed a straightforward technique to determine the ethnic origin of stem cells.
The Scripps Research scientists initiated the study—published in the January 2010 edition of the prestigious journal Nature Methods—because the availability of genetically diverse cell lines for cell replacement therapy and drug development could have important medical consequences. Research has shown that discordance between the ethnic origin of organ donors and recipients can influence medical outcomes for tissue transplantation, and that the safety and effectiveness of specific drugs can vary widely depending on ethnic background.
The team's analysis of a variety of human embryonic stem cell lines currently in use in research laboratories around the world found that these cells originated largely from Caucasian and East Asian populations, with little representation from populations originating in Africa. In response to these results, the scientists used skin cells from an individual of West African Yoruba heritage to create a new stem cell line, the first to carry the genetic profile of this ethnic group.
"Ethnic origin is a critical piece of information that should come with every cell line," said Scripps Research Professor Jeanne Loring, who is senior author of the paper. "Everyone who works with stem cells should be doing this kind of analysis."
"Knowing that a big push in the future is using these lines in the clinic and in drug development, there's a need to have an ethnically diverse population of cells," added Louise Laurent, assistant professor at the University of California, San Diego (UCSD) and research associate at Scripps Research, who is first author of the paper with Caroline Nievergelt, also an assistant professor at UCSD.
Greater diversity in cell samples would set the stage for more broadly relevant research by labs in academia and industry, more robust results on the safety and efficacy of potential therapies, and more successful tissue transplants.
The Promise of Stem Cells
Normally, cells develop from stem cells into a myriad of increasingly more specialized cell types during early development and throughout a lifetime. In humans and other mammals, these developmental events are usually irreversible. This means that when tissues are damaged or cells are lost, the body has limited means by which to replenish them.
Having a source of stem cells would be useful in many medical situations because these cells are "pluripotent," having the ability to become any of the body's cell types. Pluripotent stem cells would potentially provide physicians with the ability to replace or repair damaged tissues throughout the body. For example, pluripotent stem cells could be differentiated into the damaged cell type and transplanted.
Much research on pluripotent stem cells to date has been conducted on human embryonic stem cells, which are harvested from discarded embryos (those created but not used for the purposes of in vitro fertilization, a technique to help couples conceive). However, recently another source of pluripotent stem cells has come onto the scene. These cells—called induced pluripotent stem cells—are created by taking a sample of skin cells or another type of differentiated cell and using chemicals and molecular biology techniques to coax them back into a pluripotent state.
The current analysis included 47 human embryonic stem cell lines collected from labs located around the world—including Korea, Australia, and Finland. The analysis also included five induced pluripotent stem cell lines.
Ancestors Forgotten and Remembered
To determine the ethnic origins of the stem cell lines and to link them to genetic "signatures" that might affect medical outcomes, the scientists drew on previous research from the International HapMap Project, published in the journal Nature in 2003. This research linked single-letter alterations in the genetic code—known as single nucleotide polymorphisms, or SNPs—with people of known ethnic origins. This data provided a way to identify the ethnic heritage of a donor of any cell.
Laurent noted that simply asking cell donors about their ethnic heritage does not provide accurate data. "There's often an ancestor from a different area who a person doesn't know about," she said.
The technology used for the new study, known as SNP genotyping, uses microarrays, which are easily available, inexpensive, and relatively straight forward for scientists to use.
When the Scripps Research scientists applied the technique to the embryonic stem cell lines, they found that Caucasians were especially well represented among the samples, followed by East Asians. Cells of some mixed heritage were also common. Notably lacking from the samples were cell lines representing African heritage.
In addition, the authors found that the country in which a cell line was generated did not necessarily predict the ethnicity of the donor.
In creating a new pluripotent stem cell line from an individual with a West African Yoruba background, the scientists generated a line that contains distinct genetic markers for disease risk and drug metabolism.
"There's not a lot of value in making a new pluripotent stem cell line now unless it has something new to offer," said Loring. "I think that increasing ethnicity and genetic diversity is an important reason for generating new lines."
The data generated by the study—which Loring describes as the foundation of a new database of human pluripotent stem cell genetic information—will be available for other researchers to access for studies on specific genes, stem cell transplantation, and other topics.
In addition to Loring, Laurent, and Nievergelt, authors of the Nature Methods paper, "Restricted Ethnic Diversity in Human Embryonic Stem Cells," are: Candace Lynch and Eyitayo Fakunle of Scripps Reserach; Julie Harness of the University of California, Irvine; Uli Schmidt of Sydney IVF (Australia); Vasiliy Galat of Northwestern University Feinberg School of Medicine; Andrew Laslett of Commonwealth Scientific and Industrial Research Organisation Molecular and Health Technologies, Australian Stem Cell Centre, and Monash University; Timo Otonkoski of Biomedicum Stem Cell Center and Children's Hospital, University of Helsinki (Finland); Hans Keirstead of the University of California, Irvine; Andrew Schork of UCSD; and Hyun-Sook Park of Modern Cell and Tissue Technologies, Inc. (Korea). For more information, see http://www.nature.com/nmeth/journal/v7/n1/index.html.
The research was funded by grants from the California Institute for Regenerative Medicine, the National Institutes of Health, and the Bill and Melinda Gates Foundation.
Send comments to: mikaono[at]scripps.edu
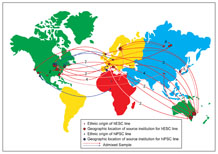
In the new study, the scientists found the ethnic origins of 52 stem cell lines. Click to enlarge.
