

Two New Antibodies Found to Cripple HIV
Researchers at and associated with The Scripps Research Institute, the International AIDS Vaccine Initiative (IAVI), and the biotechnology companies Theraclone Sciences and Monogram Biosciences have discovered two powerful new antibodies to HIV that reveal what may be an Achilles' heel on the virus.
The researchers, led by Dennis Burton, a Scripps Research professor and scientific director of the IAVI Neutralizing Antibody Center at Scripps Research, published their work on Thursday, September 3, 2009, in Science Express, an advance, online issue of the journal Science. Researchers will now try to exploit the newfound vulnerability on the virus to craft novel approaches to designing an AIDS vaccine.
"The findings themselves are an exciting advance toward the goal of an effective AIDS vaccine because now we've got a new, potentially better target on HIV to focus our efforts for vaccine design," said Wayne Koff, senior vice president of research and development at IAVI. "And having identified this one, we're set up to find more, which should further accelerate global efforts in AIDS vaccine development."
Broadly neutralizing antibodies to HIV are produced by a minority of HIV-infected individuals and are distinct from other antibodies to HIV in that they neutralize a high percentage of the many types of HIV in circulation worldwide. It is widely believed that to prevent HIV infection an AIDS vaccine would need to teach the body to produce these powerful antibodies before exposure to the virus. Animal experiments suggest that such a vaccine would work. Before this finding only four antibodies to HIV had been discovered that were widely agreed to be broadly neutralizing.
The two newly discovered broadly neutralizing antibodies, called PG9 and PG16, are the first to have been identified in more than a decade and are the first to have been isolated from donors in developing countries, where the majority of new HIV infections occur. Moreover, previously identified broadly neutralizing antibodies against HIV have functioned by binding to places on HIV that have proven difficult to exploit by means of vaccine design.
"These new antibodies, which are more potent than other antibodies described to date while maintaining great breadth, attach to a novel, and potentially more accessible site on HIV to facilitate vaccine design," said Burton, who is also a member of the newly established Ragon Institute of Massachusetts General Hospital, Massachusetts Institute of Technology, and Harvard University. "So now we may have a better chance of designing a vaccine that will elicit such broadly neutralizing antibodies, which we think are key to successful vaccine development."
Breadth of neutralization is important because any effective AIDS vaccine must provide protection from a diverse range of the most prevalent types of HIV circulating worldwide. High potency suggests that such antibodies will not have to be produced by the body in very large quantities to confer protection.
The two new antibodies target a region of the viral spike used by HIV to infect cells. The viral spike glycoproteins, termed gp120 and gp41, are highly variable and have evolved to thwart immune attack. But biochemical studies suggest that PG9 and PG16 target regions of gp120 that do not change, which probably accounts for their breadth of neutralization. Now researchers at the IAVI-organized Neutralizing Antibody Consortium (NAC), a scientific network focused on designing vaccines capable of eliciting broadly neutralizing antibodies, will turn their attention to studying the molecular structure of PG9 and PG16 and that of the region they target on the HIV spike. They will use this information to try to devise immunogens—the active ingredients of vaccines—that elicit similar antibodies.
How They Were Discovered
The methods by which PG9 and PG16 were isolated are themselves proving instructive. Their identification represents the first success of an ongoing global hunt launched by IAVI in 2006 to find new broadly neutralizing antibodies to support the rational design of novel AIDS vaccine candidates. The effort, named Protocol G, is unprecedented in scale and distinguished by its emphasis on identifying antibodies that neutralize subtypes of HIV circulating primarily in developing countries. IAVI's clinical research partners have collected blood specimens from upward of 1,800 HIV-infected volunteers from IAVI-supported clinical research centers in seven sub-Saharan countries as well as from centers in Thailand, Australia, the United Kingdom, and the United States.
All samples were sent to Monogram Biosciences, which, working with researchers at IAVI's AIDS Vaccine Design and Development Laboratory in New York City and the IAVI Neutralizing Antibody Center at Scripps Research, screened the sera for broadly neutralizing activity. Researchers historically have sought broadly neutralizing antibodies in serum by testing whether antibodies from such samples bind to soluble versions of gp120 and gp41. It turns out that PG9 and PG16, however, bind to soluble forms of the proteins very weakly, if at all. The antibodies were detected only because a micro-neutralization assay developed by Monogram in partnership with IAVI measuring their ability to block HIV infection of target cells was run in parallel with the standard binding assays used for screening. This has significant implications for the future screening of broadly neutralizing antibodies.
"If you think of it as a fishing expedition," said Christos Petropoulos, chief scientific officer and vice president of virology research and development at Monogram Biosciences, "we and the rest of the field were previously using the wrong bait in the search for HIV-specific broadly neutralizing antibodies. Together with colleagues at IAVI, we reasoned that the best approach to identifying antibodies with the most potent and broad neutralizing activity was to screen directly for their ability to block HIV infection. To do this we developed a new, specialized test known as the micro-neutralization assay, which has opened up new avenues for exploration of additional donors for similar antibodies."
Once the researchers had ranked the top 10 percent of serum samples in terms of breadth of neutralization, they needed to isolate the actual broadly neutralizing antibodies. This can be painstaking work. But Theraclone Sciences, a company that had been working outside the HIV field, had a relevant and unique high-throughput process that it adapted to HIV work with financing from IAVI's Innovation Fund, which is co-funded by the Bill & Melinda Gates Foundation. The Theraclone team used a system designed to expose the entire repertoire of antibodies from a blood sample obtained from an HIV-infected individual. Antibodies with broadly neutralizing potential were identified from this pool and traced to their corresponding antibody-forming cells. Using recombinant DNA technology, broadly neutralizing antibody genes were then isolated from these cells to enable the production of unlimited quantities of the antibody clones for research.
"It is exciting that we were able to use our technology to identify and isolate these new broadly neutralizing antibodies, which may offer important clues that could help create an effective AIDS vaccine. Through this strong scientific partnership, we have rapidly delivered promising results," said Matthew Moyle, chief scientific officer and senior vice president of Theraclone Sciences. "This project has been a useful demonstration of Theraclone's antibody discovery platform in infectious disease, and we highly value IAVI's collaborative approach to solving the AIDS vaccine challenge," said David Fanning, president and CEO of Theraclone Sciences.
With a large pool of HIV-positive donors from Protocol G now identified whose serum contains HIV-specific broadly neutralizing antibodies, it is likely that this global collaboration will generate more broadly neutralizing antibodies that will benefit the vital enterprise of accelerating AIDS vaccine development.
"The story of the discovery of these two new antibodies demonstrates the challenges of AIDS vaccine research but also the power of the collaboration that formed to produce this advance. This is what can happen when you have researchers from the global North and South, from academia and industry, from within and outside the HIV field, working together in a framework to speed innovation," said Seth Berkley, president and CEO of IAVI. "By working in this manner, I am confident we will continue to move toward solving the AIDS vaccine challenge, one of the greatest scientific and public health challenges of our time."
The first authors of the paper, "Broad and Potent Neutralizing Antibodies from an African Donor Reveal a New HIV-1 Vaccine Target," are Laura M. Walker of Scripps Research and the IAVI Neutralizing Antibody Center and Sanjay K. Phogat of IAVI's Design Lab. In addition to Burton, Walker, and Phogat, other authors include: Po-Ying Chan-Hui, Jennifer L. Mitcham, Ole A. Olsen, Steven M. Frey, Phillip W. Hammond, and Matthew Moyle of Theraclone Sciences; Denise Wagner, Stephen Kaminsky, and Timothy Zamb of IAVI's Design Lab; Melissa D. Simek, Jennifer K. Lehrman, Frances H. Priddy, and Wayne C. Koff of IAVI; Pham Phung, Julie L. Goss, and Terri Wrin of Monogram Biosciences, Inc.; Steven Fling and Pascal Poignard of Scripps Research and the IAVI Neutralizing Antibody Center; and protocol G principal investigators, who are listed as G. Miiro, J. Serwanga, A. Pozniak, D. McPhee, O. Manigart, L. Mwananyanda, E. Karita, A. Inwoley, W. Jaoko, J. DeHovitz, L.G. Bekker, P. Pitisuttithum, R. Paris, and S. Allen.
Protocol G collaborating institutions include: MRC/UVRI Uganda Research Unit on AIDS, Uganda Virus Research Institute, Entebbe, Uganda; St. Stephen's AIDS Trust, Chelsea and Westminster NHS Foundation Trust, London UK; NRL, St. Vincent's Institute, Melbourne, Victoria, Australia; Zambia Emory HIV Research Project, Lusaka, Zambia, and the Rwanda-Zambia HIV Research Group, Emory University, Atlanta, GA, USA; Projet San Francisco, Kigali, Rwanda and the Rwanda-Zambia HIV Research Group, Emory University, Atlanta, GA, USA; CeDReS/CHU Treichville, Abidjan, Côte d'Ivoire; Kenya AIDS Vaccine Initiative, College of Health Sciences, University of Nairobi, Nairobi, Kenya; SUNY Downstate Medical Center, Brooklyn, NY, USA; Desmond Tutu HIV Centre, University of Cape Town, Cape Town, South Africa; Faculty of Tropical Medicine, Mahidol University, Bangkok, Thailand; Department of Retrovirology, Armed Forces Research Institute of Medical Sciences, Bangkok, Thailand; Rwanda-Zambia HIV Research Group, Emory University, Atlanta, GA, USA; Institute of Human Virology, Plateau State Human Virology Research Centre, Jos, Nigeria.
For more information on paper, see http://www.sciencemag.org/cgi/content/abstract/1178746.
This work was supported by IAVI, IAVI's Innovation Fund (cofunded by IAVI and the Bill & Melinda Gates Foundation), the United States Agency for International Development (USAID), and the National Institute of Allergy and Infectious Diseases of the National Institutes of Health.
Send comments to: mikaono[at]scripps.edu
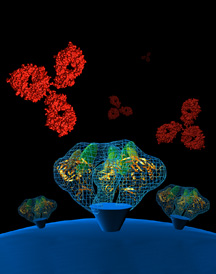
Two newly discovered broadly
neutralizing antibodies, called PG9 and PG16, are the first to have been
identified in more than a decade. Here, neutralizing antibodies (red) are
shown heading toward the viral spike (blue), where they will target
conserved regions on two loops (yellow and green). Click to enlarge. Image
by Christina Corbaci and Rob Pejchal.
