

New Study Solves Structural Mystery of Cellular Protein Transport
By Eric Sauter
Scientists at The Scripps Research Institute have determined the structure of the protective protein coat surrounding intracellular vesicles or sacs that are needed to transport proteins out of the cell. Understanding this structure could shed critically needed light on many loss-of-function diseases such as atherosclerosis, diabetes, obesity and cystic fibrosis and lead to potential new therapies.
The study was published in the August 8, 2008 edition (Volume 134, Issue 3) of the journal Cell.
The study shows a unique COPII coat complex with the ability to expand to accommodate the transport of a variety of different sized protein cargo from the endoplasmic reticulum, the convoluted network of tubes within the cell responsible for folding and transport of proteins that are delivered to the cell surface and to plasma.
"This COPII transport machinery is critical for the normal functioning of nearly one-third of proteins encoded by the genome," said Bill Balch, a Scripps Research scientist and professor of cell biology who was a senior author of the syudy. "So, any structural anomaly or improper engagement of this coat machinery can result in a number of debilitating and chronic diseases. Finally understanding this structure opens the door to the possibility of developing advanced therapeutic solutions."
The study, conducted in collaboration with Scripps Research scientists Bridget Carragher and Clint Potter also of the National Resource for Automated Molecular Microscopy (NRAMM), shows for the first time the adaptive layer responsible for recognizing protein cargo. This interacts with an outer cage lattice that contains an unanticipated flexible hinge region that can direct the expansion of the cage that surrounds the vesicle to hold large molecules such as bone precursors and cholesterol rich fat particles that are particularly sensitive to this function. Inherited malfunctions of this mechanism result in a number of diseases.
"Before these findings, the general consensus was that the transport machine basically ran constantly and that the protein cargo somehow got incorporated into it," Balch said. "But no one had actually ever seen the structure generated by the Carragher and Potter groups as an intact, assembled coat. Our study suggests a new mechanism by which the cargo drives coat assembly for vesicle transport from the endoplasmic reticulum." When this common mechanism fails in many diseases, human health is put at risk.
The Export Business of the Cell
The transport of proteins is one of the ways that cells in various body tissues maintain specialized functions. A complicated series of events involving hundreds of different molecular components gather together proteins for export by folding and packaging them in anticipation of shipping to their final destination.
However, the study finds, the structural relationships between the protein cargo, the adaptor complexes, and cage components that allow the creation of vesicles at endoplasmic reticulum exit sites have not been well defined.
"Think of it in terms of a car," Balch suggested. "Before our study, looking at the transport machine was like looking at a pile of car parts--you had all the parts, but you couldn't tell what was going on or what would happen next. Now we have a fully assembled car. We know the structure and we can finally begin working on it to see precisely how it functions."
In studying the newly uncovered structure, Balch and his colleagues found that the COPII adaptor layer itself self-assembles into lattice-like clusters in a unique and unanticipated way.
"Working with the Carragher and Potter groups, we generated a 3-D model of a large icosidodecahedral cage structure--a polyhedron with 12 pentagon shaped areas and 20 triangular shaped areas--that contains the adaptor layer," Balch said. "As it turns out, sections of the adaptor layer are assembled in an off-center or asymmetric fashion, which contributes significantly to the mechanism by which the cage lattice architecture is generated, allowing a considerable flexibility that facilitates cage expansion."
The study also confirms that the protein cargo is in charge of cage assembly.
"To construct the cage, the adaptor complex has to preassemble, but the only way it can do that asymmetrically is if the protein cargo is at the heart of the system," Balch said. "If the protein cargo has a problem, a misfold for example, it can't recruit the adaptor legitimately to drive cage assembly. This is what happens in disease."
This implies that in protein misfolding diseases such as cystic fibrosis, the critical issue is more than simply the misfolded protein.
"The reality is that these mutations disconnect the protein cargo from the adaptor system," Balch said. "In misfolding diseases, the mutation destabilizes the protein folding and the cargo can't carry out the assembly of the adaptor complex. That tells us that there's a common basis for this adaptor complex that is crucial for every one of these diseases."
Cage Transport
In cystic fibrosis, Balch said, because the cargo fails to recognize the transport machinery, the proteins are ultimately degraded. The cell keeps producing proteins, but the proteins can't make it past the steps needed to build the right kind of cage assembly. When enough of these proteins fail to reach their correct position on the surface of cells in the lung, pancreas, and intestine, the mucus on the cell surface in these tissues becomes thick and sticky, setting off a cascade of events that is cystic fibrosis.
"This shows just how important assembling an adaptor cluster is, and in these diseases we have a failure to drive this function," he said. "It's not just misfolding. Other diseases face the same problem. Hence we have identified a critical common event in the life of a cell that contributes significantly to human health."
The structure of the vesicle coat containing the adaptor layer in the study should provide a working model for the assembly of adaptors in other coat structures such as COPI and clathrin, Balch added. "Right now, we're trying to understand exactly how it assembles," he said. "Because this is a self-assembling nano particle, we believe there must be proteins that ultimately regulate the assembly."
Other authors of the study, Structural Basis for Cargo Regulation of COPII Coat Assembly, include Paul LaPointe, Abbas Razvi, Clinton S. Potter, and Bridget Carragher of The Scripps Research Institute; Cemal Gürkan of The Scripps Research Institute and The Cyprus Institute of Neurology and Genetics; and Scott M. Stagg of The Scripps Research Institute and Florida State University. See http://www.cell.com/content/article/abstract?uid=PIIS0092867408007769.
The study was supported by the National Institutes of Health, and the Cystic Fibrosis Foundation.
Send comments to: mikaono[at]scripps.edu
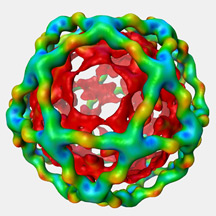
Scripps Research Institute scientists have determined the structure of a vesicle coat that transports proteins involved in diverse diseases including diabetes and cystic fibrosis, providing key insight into the origins of pathology. Image by Mike Pique.
