

A New Slap Against Malaria
By Ramya TN Chakravarthy
I know this little thing
A myriad men will save.
O Death, where is thy sting?
Thy victory, O Grave?
Inspired by his discovery that mosquitoes, and not "bad air," carry the malaria parasite, Plasmodium, a hopeful Ronald Ross penned these lines in 1897. The use of insecticides, a direct fallout of Ross's research, helped control malaria in the following years.
Malaria has, however, lately re-emerged as a killer disease, largely due to widespread resistance to commonly used antimalarial drugs. More than a century after Ross's pioneering research, scientists at The Scripps Research Institute might have found another way to knock the sting off this dreaded disease.
Cashing in on Calcium
In research published in an advanced, online edition of Nature Chemical Biology this month, a team of scientists led by Scripps Research Associate Professor Elizabeth Winzeler focused on illuminating the role of a calcium sensing protein kinase, PfCDPK1, expressed in the malaria parasite, Plasmodium falciparum.
Plasmodium is an intracellular parasite that invades red blood cells, and scientists have known that calcium is essential for parasite invasion. The scientists reasoned that PfCDPK1 might somehow be connected with the parasite's invasion of host red blood cells, part of its complex lifecycle.
"We were wondering what PfCDPK1 might be doing," says Winzeler. "We looked at where it was expressed and it appeared to be coincident with a lot of genes involved in parasite motility."
It was a clear case of guilt by association.
The team conducted a series of biochemical experiments that revealed that PfCDPK1 was not only involved in parasite motor functions, but was also essential for parasite survival. This meant that the protein could potentially be an exciting and novel target for the development of antimalarial drugs.
A Promising Candidate
The scientists developed this idea further by testing the protein against a chemical library of 20,000 compounds. They found that a class of compounds, including one they named "purfalcamine," was capable of inhibiting PfCDPK1 activity. Purfalcamine inhibited parasite motility, leading to the arrest of parasite growth and eventually killing the parasites.
While this type of compound arrested Plasmodium growth in vitro, the compounds tested were not potent enough in their current form to completely kill malaria parasites in vivo. That might change, however, with the application of rational drug design.
The paper represents a promising step in the direction of a new therapy for malaria, and would not have been possible without the contributions of scientists with diverse specialties. Winzeler notes, "It was great working on such a collaborative project with scientists from different fields—biologists, chemists, bioinformaticians…"
In addition to Winzeler, the research team includes N. Kato, G. Breton, K. Henson, J. Johnson, F. Marr, V. Ramachandran, and P. G. Schultz from The Scripps Research Institute; T. Sakata, K. G. Le Roch, A. Nagle, C. Andersen, B. Bursulaya, D. Mason, C. McNamara, D. Plouffe, M. Spooner, T. Tuntland, Y. Zhou, E. C. Peters, A. Chatterjee, and N. Gray from the Genomics Institute of the Novartis Research Foundation; K A Kumar from the New York University School of Medicine; G. E. Ward from the University of Vermont; and J. Harper from the University of Nevada.
The project was funded by grants to Winzeler from the Ellison Foundation, the Keck Foundation, and the U.S. National Institutes of Health, a grant to Ward from the U.S. National Institutes of Health, and grants to the Genomics Institute of the Novartis Research Foundation from the Wellcome Trust and the Medicines for Malaria Venture.
For more information, see the Nature Chemical Biology website at: http://www.nature.com/nchembio/journal/vaop/ncurrent/abs/nchembio.87.html.
Send comments to: mikaono[at]scripps.edu
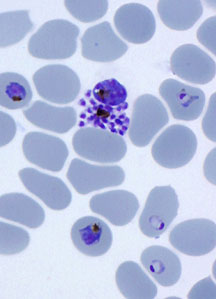
A team led by scientist Elizabeth Winzeler found that a calcium sensing protein kinase, PfCDPK1, is essential for the survival of malaria parasites, like these shown here (dark purple) infecting red blood cells (lighter purple).
