

Team Identifies Human Antibodies that Prevent Hepatitis C Virus Infection in Mouse Model
By Mika Ono
A team of researchers has found that certain antibodies can prevent hepatitis C virus infection in a "humanized" mouse model, opening the door to the use of antibodies as a human therapeutic and the development of a preventive vaccine for the disease.
The work was published in the advance, online edition of the journal Nature Medicine on December 6, 2007.
The study's authors, who include Scripps Research Professor Dennis Burton, University of Alberta Professor Norman Kneteman, and an international team of colleagues, identified a group of special antibodies that can broadly neutralize hepatitis C virus. Antibodies (proteins produced by the body's immune system in response to a foreign substance) are considered to be broadly neutralizing when they are effective against many different strains of a pathogen.
"The findings that neutralizing human antibodies attack a region commonly found on the surface of many hepatitis C virus strains and [that these antibodies] protect against infection by the virus in an animal model provide proof of principle for the viability of passive immunotherapy against this extremely variable virus," says Mansun Law, a research associate at Scripps Research who was first author of the paper. "The results also raise hopes for the development of an effective vaccine to prevent the disease from taking hold."
Preventing Hepatitis C Virus Infection
Hepatitis C is a potentially fatal disease of the liver caused by the hepatitis C virus, which is spread by contact with the blood of an infected person. Those at highest risk include people with a history of injecting illegal drugs, participating in unsafe sexual practices, receiving a blood transfusion or solid organ transplant before 1992, or being on long-term kidney dialysis. According to the Centers for Disease Control and Prevention (CDC) and the World Health Organization (WHO), an estimated 4.1 million Americans have been infected with the virus and two to three percent of the world's population is chronically infected.
The course of the disease varies. A small fraction of people contracting the virus develop acute viral infection but produce a strong enough immune response to clear it. Most people contracting the virus, however, become chronically infected without being aware they have the disease. Chronic infection can result in loss of liver function, cirrhosis (scarring of the liver), and liver cancer.
While treatment of chronic hepatitis C infection with a combination therapy of pegylated interferon and the anti-viral drug ribavirin is successful about half the time, hepatitis C remains the leading cause of liver cancer and the leading indication for liver transplants in the United States.
"There is an urgent need to discover means to prevent new infection," said Law, "and to treat those infected [more effectively]."
Currently, no vaccine exists to prevent hepatitis C. A major problem in developing vaccines has been the virus's extreme variability. For a vaccine to be practical, it must evoke antibodies that counteract many of the strains of the hepatitis C virus circulating in the human population, not only a small subset.
In the current Nature Medicine study, for the first time scientists were able to identify just such antibodies, define the regions on the virus that these powerful antibodies target, and demonstrate their activity in animals.
The researchers began the study by isolating a panel of antibodies from a donor chronically infected with hepatitis C virus. Using cell culture-based assays, the researchers challenged each of these antibodies with a variety of strains of hepatitis C virus to see which, if any, were effective across the board.
The results showed that broadly neutralizing antibodies do exist, and that they target a part of the hepatitis C virus that the researchers called antigenic region 3. The other antibodies, which the researchers isolated more frequently, failed to neutralize as many different strains and targeted other parts of the virus, antigenic regions 1 and 2.
Law notes that antigenic region 3 is a part of the virus that is "relatively conserved," in other words unchanged despite many other mutations that distinguish among different strains. Often, a region of a virus is conserved because it is necessary for the virus's survival, for example, essential for the ability to reproduce.
In the next part of the study, the researchers tested the antibodies that were broadly neutralizing in cell culture in a small animal model designed to mimic human response—mice with livers containing human liver cells. One group of these mice received an injection of broadly neutralizing antibodies, while a control group did not. Both groups were then exposed to a high dose of hepatitis C virus quasispecies from another infected donor. Virus quasispecies, or a population of closely related virus strains, are produced during an infection by viruses that have an exceptionally high replication and mutation rate like hepatitis C virus. The production of virus quasispecies is a powerful mechanism for viruses to escape immune surveillance and anti-viral drugs.
The mice who had received the broadly neutralizing antibodies were either fully protected from contracting the disease ("sterilizing protection") or protected until levels of the antibody decayed. All animals in the control group were infected and unable to prevent the virus from proliferating.
Toward Vaccines and Therapies
The researchers plan to follow up on these results, pursuing potential vaccine candidates, developing antibodies that might be administered directly as a therapy in a technique called "passive immunization," and laying groundwork for future drug development.
On the vaccine front, the scientists will investigate how to induce the body to produce the specific type of broadly neutralizing antibodies discovered in the recent study. On the passive immunization front, the researchers will see if they can increase the antibodies' anti-viral activity. In addition, the team will seek to identify additional conserved targets on the virus, which could prove useful for vaccine, antibody, and drug development.
"In order to cure highly variable viruses, such as hepatitis C virus and HIV," said Law, "it will probably be necessary to use a combination of highly potent antibodies and anti-viral molecules to attack multiple conserved regions in the virus."
Law believes that antibody administration alone would be most useful in cases in which a health care worker has been accidentally stuck with an infected needle, or after a liver transplant, to prevent re-infection of the new organ.
In addition to Law, Burton, and Kneteman, authors of the new study, "Broadly Neutralizing Antibodies Protect Against Hepatitis C Virus Quasispecies Challenge," are Jamie Lewis of the University of Alberta, Canada; Alexander W. Tarr and Jonathan K. Ball of the University of Nottingham, Queen's Medical Centre, U.K.; Zania Stamataki and Jane A. McKeating of the University of Birmingham, U.K.; Ian M. Jones of the University of Reading, U.K.; Robert I. Fox of Scripps Memorial Hospital; and Toshiaki Maruyama, Erick Giang, Pablo Gastaminza, and Francis V. Chisari of The Scripps Research Institute. The study was not supported by specific funding agencies, but partial salary support was provided by the Elizabeth Glaser Pediatric AIDS Foundation, the Sjögren's Syndrome Foundation, the Arthritis Foundation, the Alberta Heritage Foundation for Medical Research, and a Wyeth Canada–Canadian Institutes for Health Research Clinical Research Chair in Transplantation.
For more information on the study, see Nature Medicine at http://www.nature.com/nm/journal/vaop/ncurrent/abs/nm1698.html.
Send comments to: mikaono[at]scripps.edu
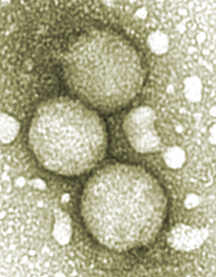
"There is an urgent need to discover means to prevent new infection [from hepatitis C virus] and to treat those infected [more effectively]," says Mansun Law of the Burton lab, an author of the new Nature Medicine paper. Image of particles isolated from hepatitis C virus-infected cell culture provided by Pablo Gastaminza and Francis V. Chisari. Click for scientific details.
