

Scripps Research Team Unravels Drug Target for Parasitic Diseases
By Mark Schrope
The ongoing search for better treatments for devastating parasitic diseases such as Chagas' disease and African sleeping sickness now has a new target, thanks to research by a team from The Scripps Research Institute. The group now understands better a critical DNA-protein binding event that, if blocked, can kill the parasites that cause the diseases. The researchers are already working to screen drugs that will block this mechanism.
"I think we now understand far more about, arguably, the best pharmaceutical target out there right now for treating some of these diseases," said Scripps Research chemist Paul Wentworth, Jr., who led the study appearing in the cover article of the April 13, 2007 issue of Angewandte Chemie.
The family of diseases the Scripps Research group is targeting is caused by parasites known as kinetoplastids, which include Trypanosoma and Leishmania afflicting millions globally, mainly in developing countries. The diseases, which are transmitted by flies, include Chagas' disease, a condition common in Central and South America, and East and West African sleeping sicknesses. They result in painful, long-lasting lesions or potentially fatal problems with internal organs. "[These types of illnesses] are really are a global menace and truly terrible diseases," said Wentworth. The diseases are of increasing interest in the United States due to the growing number of soldiers who are exposed during duty in areas such as Afghanistan.
Current treatments are less than ideal because they can be toxic to humans, and because the parasites responsible are growing increasingly resistant to them. This means the many academic and commercial groups working on new treatments are in need of new disease targets to pursue.
In the 1990s, Piet Borst of the University of Amsterdam and the Netherlands Cancer Institute discovered that these parasites have a unique modification in their DNA not seen in any other type of organisms. The modification involves the addition of a glucose molecule to portions of DNA, namely the pyrimidine base of uracil nucleosides. Borst and his team, which collaborates with Scripps Research investigators, later discovered that the parasites have a protein, dubbed JBP1, that binds specifically to this modified base, known as nucleoside dJ, or Base J.
The exact function of the protein is not yet known, but studies by Borst and his team have shown that genetic alterations that block production of the protein kill the parasites. This strongly suggests that blocking the protein from binding to DNA would also kill the parasites. "We got involved to ask the question, 'How would you even begin to do this?'" said Wentworth.
As a first step toward answering that question, Wentworth and his group synthesized a panel of Base J analogs and incorporated them into double-stranded DNA. Scripps Research scientist David Millar then studied the binding of these oligonucleotides containing base J analogs to the JBP1 protein, which was produced by Prem Subramaniam, a postdoctoral researcher in Charles Weissman's group at Scripps Florida. Fluorescence anistropy was then used to measure how well the protein was binding to a given altered molecule.
The group was surprised to discover that there was no clear pattern in how differing alterations to base J affected binding to JBP1. To help solve the puzzle, David Case, another Scripps Research investigator, created computer models of the various double-stranded DNA sequences containing the different base J analogs. By analyzing the binding data along with the models, the group was able to deduce that the most critical component of the mechanism was how the glucose initially binds to the backbone in a specific groove of the DNA molecule. The alterations that were blocking binding were those that either prevented the glucose from lining up properly in the groove to expose the portion with which JBP1 binds, or blocking off the groove altogether.
The protein can only bind to glucose properly oriented in the DNA groove, meaning that a drug treatment that alters or blocks the groove should kill the parasites. The team is already working to screen libraries of such compounds in the hopes of identifying drugs that have one or the other of these effects.
Wentworth cautions that the work is largely theoretical, because the molecules involved have not been imaged, so the orientations of the molecules thought to be important can only be inferred. However, work is ongoing in other Scripps Research labs to resolve the structures of the components involved using x-ray crystallography. This could confirm the theories as well as help to identify additional potential drug targets.
The paper is titled "O-Glycoside Orientation Is an Essential Aspect of Base J Recognition by the Kinetoplastid DNA-Binding Protein JBP1." In addition to Wentworth, Millar, and Case, other authors on the paper were Rajesh Grover, Stephanie Pond, Qizhi Cui, and Prem Subramaniam, all from Scripps Research. See http://www3.interscience.wiley.com/cgi-bin/abstract/114121534/ABSTRACT.
Send comments to: mikaono[at]scripps.edu
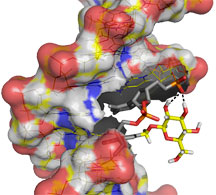
A cover article in Angewandte Chemie illuminates a critical DNA-protein binding event, providing a new target in the fight against devastating parasitic diseases. Click for details.
