

New Class of Enzyme Inhibitors Block Replication of SARS Virus
By Eric Niiler
Scientists at The Scripps Research Institute have discovered a class of compounds that block the SARS virus from replicating, a finding that may open the door to new drug targets against the deadly disease.
The study was conducted by researchers from Scripps Research; the Genomics Research Center, Academia Sinica, Taiwan; and the National Taiwan University. It is being published in an advance online version of the journal Chemistry and Biology.
Chi-Huey Wong is currently the Ernest W. Hahn Chair in Chemistry at the Skaggs Institute of Chemical Biology and directs the Scripps Research lab heading the study. He said the new finding is an important step in developing a possible drug treatment against SARS.
"We have been working on the problem of SARS since the epidemic started in 2003," Wong said. "This new class of inhibitors represents the most potent SARS virus protease inhibitors known today."
The path to the new research finding has taken several years. In 2002, Severe Acute Respiratory Syndrome (SARS) emerged in rural China and eventually spread to 32 countries, according to the World Health Organization. SARS is caused by a ring-shaped virus, known as a coronavirus. The SARS coronavirus is suspected of originating in animal populations before migrating to humans. Hardest-hit were six Asian nations. By the time the epidemic had been controlled in 2003, the disease infected more than 8,000 people, causing 800 deaths. There is no current effective treatment or vaccine.
Researchers have known since 2003 that a site on the virus is responsible for mediating proteases that allow the virus to replicate. Since then researchers have been testing protease inhibitors to lock up this site, known as SARS 3CLpro, and effectively stop the virus from infecting additional cells in the body.
In 2004, Wong's lab discovered that Lopinavir, a protease inhibitor of HIV also known as TL3, also served as weak inhibitor of the SARS 3CLpro site (PNAS,101, 10012-10017). Since then, members of Wong's group further studied Lopinavir and are preparing it for clinical trials against SARS.
Researchers in Wong's lab at Scripps Research and in Taiwan have been looking at other Liponavir-related compounds for similar blocking effects. During these experiments, they found that a group of catalyzing agents used to helped promote chemical reactions in the laboratory were actually more powerful in blocking the SARS protease than the either Lopinavir or any of the target compounds.
These organic compounds are called benzotriazole esters. The esters entered the SARS protease site, formed an intermediary compound, then inactivated the SARS enzyme. The findings were confirmed using mass spectrometry analysis of the enzyme intermediary.
"These benzotriazole esters are relatively stable and act as suicide inhibitors," Wong said. "They block the enzyme, are transformed through a co-valent bond and are unable to get out."
Wong said the findings provide better insight into the mode of action of the enzyme, which may lead to development of a drug against SARS. The research used rapid drug discovery techniques developed in the Wong lab to screen large numbers of weak enzyme inhibitors, then attached additional compounds to look for stronger reactions.
Research Associate Chung-Yi Wu, a member of the Wong lab, is the paper's lead author. He said the finding was unexpected.
"We wanted to improve Liponavir activity," Wu said. "But we found this very surprising and serendipitous result."
Other authors of the study include Ke-Yung King, Chih-Jun Kuo, Jim-Min Fang, Ying-Ta Wu, Ming-Yi Ho, Chung-Lin Liao, Jiun-Jie Shie and Po-Huang Liang of the Genomcs Research Center, Academia Sinica, Taiwan and National Taiwan University, Taipei, Taiwan. The study was supported by the National Science Council, Taiwan, and Genomics Research Center, Academia Sinica. The work is supported by the National Science Council, Taiwan and Genomics Research Center, Academia Sinica.
Send comments to: mikaono[at]scripps.edu

"This new class of inhibitors represents the most potent SARS virus protease inhibitors known today," says Professor Chi-Huey Wong.
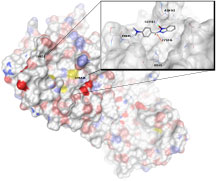
Computer modeling of benzotriazole binding to SARS-CoV 3CL protease. Click to enlarge
