A Simple Strategy for Blocking HIV Transmission Proves Effective in Pre-Clinical Trials
By Jason Socrates Bardi
An international team of researchers recently announced the promising results of a preclinical study on a chemical called PSC-RANTES to block male-to-female sexual transmission of human immunodeficiency virus (HIV). In this week's issue of the journal Science, the team is publishing a report that describes how a topical microbicide with a high enough concentration of PSC-RANTES prevented HIV transmission to female rhesus macaques that were challenged with high doses of a modified form of HIV.
The corresponding author of the report was Michael Lederman, M.D., who is Scott R. Inkley Professor of Medicine and director of the Case Western Reserve University/University Hospitals of Cleveland Center for AIDS Research, in Cleveland, Ohio. Other important contributors to the research include Robin Offord, Ph.D., and Oliver Hartley, Ph.D., of the University of Geneva in Switzerland, the chemists who designed the chemical PSC-RANTES, and Ronald Veazey, DVM, who conducted the animal studies at the Tulane National Primate Research Center.
Donald Mosier, M.D., Ph.D., who is a professor of immunology at The Scripps Research Institute, is also a member of this research team and one of the authors on the paper. Mosier has long been Lederman's collaborator on the project, and he contributed much of the preliminary work on characterizing PSC-RANTES prior to the animal studies.
PSC-RANTES works by targeting a protein in the body called C–C chemokine receptor 5 (CCR5)—the receptor on human cells to which HIV binds. HIV needs CCR5 in order to achieve infection of any given cell. In order for the virus to be transmitted during heterosexual intercourse—the number one way the virus is spread in many parts of the world—the virus must attach to CCR5 in cells within the vaginal mucosa. When these cells are protected with PSC-RANTES, however, the virus cannot attach to them.
"This molecule causes CCR5 to be internalized inside the cell and hide from the virus for long enough to prevent infection," says Mosier.
Learning From Resistance
The World Health Organization estimates that to date more than 20 million people have died from HIV and that some 40 million people are living with HIV worldwide. During 2001 alone, more than four million men, women, and children succumbed to the disease, and by the end of that year, the disease had made orphans of 14 million children. In the United States, 40,000 people are infected with HIV each year—more than one person every 15 minutes.
One of the greatest public health prerogatives facing the world today is to develop effective ways to curtail transmission. The study results that are being announced this week are a step in that direction. How large of a step? "It is a proof-of-principle that you can target CCR5, block it, and block transmission of HIV," says Mosier.
Mosier and his colleagues have studied the basic mechanism of HIV's interaction with CCR5 since it was first discovered in the mid-1990s in certain individuals who seemed to be protected from HIV infections despite having had multiple high-risk exposures to the virus. It turns out that these people have mutations in their CCR5 genes.
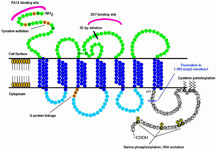
Normally, CCR5 is a receptor that helps immune cells receive external signals. It sits on the surface of helper T cells and other types of human cells—the very ones that are susceptible to infection with HIV. There, this protein of 332 amino acids spans the cell membranes of human CD4+ T helper cells, with the N-terminus and three external "binding" loops exposed to the outside and the C-terminus and three internal "signaling" loops on the inside.
Once a chemokine that binds to CCR5 comes along, which occurs during both viral and bacterial infections, it will attach to CCR5's binding loops, and this event will cause the molecule's signaling loops on the inside of the cell to mediate internalization. During internalization, both the CCR5 and the chemokine bound to it are pulled into the cell, where they are contained in endocytic vesicles. As the contents of the vesicle are acidified, the CCR5 dissociates from its bound molecule and then is transported back to the surface.
Mosier, who has helped to elucidate this basic mechanism of binding-and-internalization in the last few years, says the idea of exploring CCR5 as a possible target for intervention came after scientists realized that people with the CCR5 gene mutations are perfectly normal except that they are resistant to HIV infection.
The resistant individuals all had a 32-base pair knockout mutation in their CCR5 genes, which causes their CCR5 proteins to be truncated. This truncation prevents the CCR5 proteins from appearing on the surface of cells and acting as receptors for HIV virions. These people were resistant to HIV infection. Even individuals who were heterozygous for the mutation had lower CCR5 expression levels, less cell-to-cell infection, and brighter clinical prognoses.
"Since the virus has to use this receptor," says Mosier, "[we reasoned that] one might be able to block the interaction and prevent infection."
Over the last few years, Mosier and his colleagues have worked to design and test agents that would bind to CCR5 and prevent it from functioning as an HIV receptor—molecules like the 68-amino acid peptide RANTES. RANTES is a natural human signaling molecule that binds to CCR5. When it does, it causes the CCR5 molecules to be pulled inside the cell for about 20 minutes.
This fact made RANTES a good lead candidate, and using it as a starting point, Mosier's colleagues were able to design a form of the peptide called PSC-RANTES where the three amino acids on the N terminus are modified. PSC-RANTES is 1,000-fold more potent at blocking HIV cell entry than the natural human signaling molecule.
Normally it takes about 20 minutes for the CCR5 molecules to return to the surface of their cell after they have been internalized due to RANTES binding. However, a large dose of the PSC-RANTES can completely strip the CCR5 molecules off the cell walls and shuttle them to sites near the nuclei of the cells. Only after 24 hours will receptors again appear on the outside of the cells.
"The CCR5 goes in and does not come back out," says Mosier. "It looks like [PSC-RANTES] has a really durable effect in that one exposure takes all the CCR5 in, and it can't come back out for a day or two."
Mosier did much of the basic science work that led to the selection of PSC-RANTES as the compound that the team would move into pre-clinical trials. In these trials, they used a topical microbicide formulated with PSC-RANTES to protect rhesus macaques that received an extremely high viral challenge. Those macaques that had been given a high concentration PSC-RANTES were protected from infection with a modified, chimera form of simian immunodeficiency virus (SIV) called simian-human immunodeficiency virus (SHIV).
If, in the future, a topical microbicidecontaining PSC-RANTES proves to be effective in humans in the context of clinical trials, it would be a boon to worldwide efforts to contain and curtail the global HIV epidemic because such a microbicide would be broadly efficacious against the many HIV strains that exist in the world. These strains have arisen because HIV replicates with such notoriously low fidelity that it frequently makes mistakes. This has been the bane of HIV therapy, because even as scientists have developed drugs to treat the virus in infected patients, drug-resistant strains have emerged. A microbicidal barrier based on PSC-RANTES would have the potential to target HIV strains broadly.
"Virtually all HIV [strains] use the CCR5 receptor," says Mosier. "[So, completely] blocking CCR5 would block 99 percent of HIV transmission worldwide."
To read the article, "Prevention of Vaginal SHIV Transmission in Rhesus Macaques Through Inhibition of CCR5" by Michael M. Lederman, Ronald S. Veazey, Robin Offord, Donald E. Mosier, Jason Dufour, Megan Mefford, Michael Piatak Jr., Jeffrey D. Lifson, Janelle R. Salkowitz, Benigno Rodriguez, Andrew Blauvelt, and Oliver Hartley, please see the October 15, 2004 issue of the journal Science or go to: http://www.sciencemag.org.
This work was supported by the National Institute of Allergy and Infectious Diseases, the Swiss National Science Foundation, and the National Cancer Institute.
Send comments to: jasonb@scripps.edu

"This molecule [we've been studying] causes CCR5 to be internalized inside the cell and hide from the [HIV] virus for long enough to prevent infection," says Professor Donald Mosier.
CCR5 molecule and the general orientation in the membrane. Click to enlarge.