Antibodies and Infectious Disease
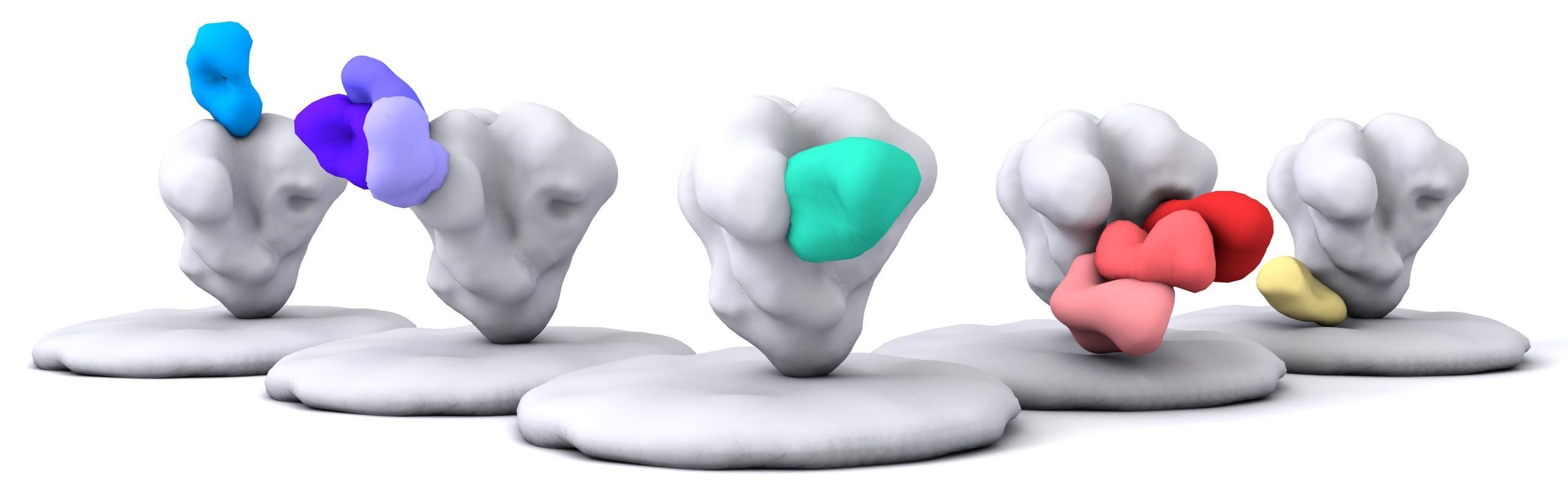
The perception of man as the easy victor over microbes has changed dramatically in the last two decades. Vaccination has offered protection against a number of viral pathogens, but it is increasingly recognized that the strategies used in the past will not be successful against all viruses. We are focused on developing rational vaccine strategies, particularly against HIV and particularly using an approach termed “Reverse Vaccinology 2.0”. In this approach, broadly neutralizing antibodies isolated from natural HIV infection are investigated in interaction with their sole viral target, the HIV Envelope, and the data used to guide immunogen design and immunization strategies. Immunogens are then evaluated in detail in animal models and the results used to iteratively improve immunogens as we move toward an HIV vaccine suitable to protect humans.
News
San Diego scientists looking for COVID-19 survivors whose blood might help fight the virus
COVID-19 survivors needed for Scripps Research antibody study
Monkey-infecting virus may provide part of future HIV vaccine
Scripps Research discoveries pave way to clinical trial of new type of HIV vaccine
Humans may be capable of producing a quintillion different antibodies
HIV vaccine protects non-human primates from infection
Scripps Research scientists receive $12 million for malaria and flu vaccine research
Scripps Research Study Reveals New Clues to How a Successful HIV Vaccine Could Work
Focus: In struggle against HIV, scientists get help from cows
NIH-supported scientists elicit broadly neutralizing antibodies to HIV in calves
San Diego Team Tests Best Delivery Mode for Potential HIV Vaccine
Team Discovers Antibodies that Target Holes in HIV's Defenses
Team Harnesses Antibody Evolution on the Path to an AIDS Vaccine
Study Supports Strategy of Series of Shots to Vaccinate Against AIDS
Researchers Discover New Ebola-Fighting Antibodies in Blood of Outbreak Survivor
Team Finds New Point of Attack on HIV for Vaccine Development
Promising HIV Vaccine Target Found
Hitting a Moving Target: AIDS Vaccine Could Work Against Changeable Site on HIV