

Scientists Develop New Technology for a Mercury Contamination Screening "Kit"
By Jason Socrates Bardi
Scientists at The Scripps Research Institute and Xenobe Research Institute have developed an improved screening method that can detect mercury contamination in biological samples including fish. At the heart of the new method is a chemical "ligand" the scientists synthesized that binds to mercury and other toxic heavy metals.
The ligand is inexpensive, easy to synthesize, and causes mercury and other toxic heavy metals to precipitate instantaneously into an easily detectable solid, says Scripps Research investigator Kim D. Janda. The method could be developed into a kit that could be used both by consumers and environmental professionals to detect metal contamination.
"The detection is very simple but very powerful," he says.
Janda is the Ely R. Callaway Chair in Chemistry and a professor in The Skaggs Institute for Chemical Biology at The Scripps Research Institute. He led the scientific team, which includes Tobin Dickerson, a recent graduate of the Scripps Research Kellogg School of Science and Technology and James La Clair, who is the director of Xenobe Research Institute in San Diego, CA.
An Invisible Toxin and a Way to Detect it
Mercury is a naturally occurring element (number 80 on the periodic table), which is usually found in only trace amounts in nature. However, according to the U.S. Environmental Protection Agency (EPA), industrialism has increased the amount of mercury in the environment.
Tons of mercury is emitted into the air every year from the burning of coal and the disposal of hazardous waste from industrial boilers and other sources. The production of chlorine also releases large amounts of mercury, and other manufacturing processes may lead to the inadvertent contamination of soil or groundwater.
Mercury in the air or soil eventually reaches the oceans and groundwater, where aquatic microorganisms have the ability to convert the elemental form of mercury into the highly poisonous methylmercury. Methylmercury in water can then accumulate in the tissues of fish and marine mammals. Fish absorb it through their gills or digestive tracts, and the poison accumulates in their tissue. Larger fish are the most likely to accumulate significant amounts because they eat smaller fish and have longer life spans.
Methylmercury in fish is a serious health hazard, especially for children and pregnant women, because methylmercury interferes with developing nervous systems. According to the U.S. Environmental Protection Agency (EPA), the most common way that people are exposed to mercury is by eating fish. Earlier this year, the EPA issued an advisory to pregnant women, women who might become pregnant, nursing mothers, and young children to avoid eating larger, high-risk fish, such as swordfish, king mackerel, and tilefish. The U.S. Food and Drug Administration (FDA), which works with state regulators and commercial fisheries to monitor methylmercury levels, also has issued a warning recommending a mercury limit of no more than 1 part per million for human consumption.
Method Detects Mercury in Much Lower Concentrations
In the latest issue of the Journal of the American Chemical Society, Dickerson, La Clair, and Janda report how they tested dozens of different aqueous solutions containing various concentrations of metal ions, and found that the ligand precipitates mercury very efficiently. In fact, it precipitated even trace amounts of the metal from solutions containing almost 10,000 times less mercury than the EPA limit. The scientists were also able to sensitively detect traces of other toxic heavy metals, such as lead and cadmium ions.
In contrast, when they tested their detection method on solutions with non-toxic metals like copper and iron, the ligand did not precipitate and they did not detect these metals. This was important because the method does not generate false positives from detecting non-toxic metals like copper and iron normally found in fish and other biological tissues.
The investigators are now comparing their method to existing assays.
If their method is comparable to existing methods for detecting mercury in fish, it could be a boon to consumers and field environmentalists who monitor toxins in the environment because it would be faster and less expensive than existing methods, which rely on laboriously catching whole fish and bringing them into a laboratory for slow, expensive, and complicated testing.
Dickerson, La Clair, and Janda have begun to adapt their method for field use by making small capillary tubes that are preloaded with the ligand. When one of these capillaries is used to stab a piece of fish, the precipitation reaction would take place and could be detected by looking at the capillary under a field detector or microscope. This method could be used to screen thousands of samples a day, the authors say.
They also envision that it would not be difficult to develop a simple consumer assay where a similar capillary could be placed in a detection device plugged into a home computer, allowing consumers to test their own fish and determine whether they are contaminated with unsafe levels of mercury for perhaps as little as a few dollars per test.
At the moment, the kit is still in development and is not available to the public.
The article, "A Precipitator for the Detection of Thiophilic Metals in Aqua," is authored by Tobin J. Dickerson, Neal N. Reed, James J. La Clair, and Kim D. Janda and will appear in an upcoming issue of the Journal of the American Chemical Society.
The research was funded by The Skaggs Institute for Research, by an Eli Lilly Graduate Fellowship, and by a Norton B. Gilula Graduate Student Fellowship.
Send comments to: jasonb@scripps.edu

Research Associate Tobin Dickerson (left) and Professor Kim Janda have developed an improved screening method that can detect mercury contamination in biological samples. Photo by Tom Gatz.
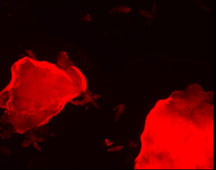
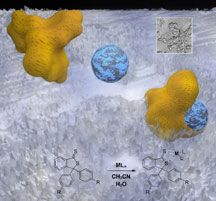
Images from the new mercury-detection technology. Click to enlarge. Courtesy of
J. La Clair.
