Give Me Immunity or Give Me Death
By Jason Socrates Bardi
Having
been among the early converts, in this part of the globe, to [smallpox
vaccine's] efficiency, I took an early part in recommending it to my
countrymen. I avail myself of this occasion of rendering you a portion
of the tribute of gratitude due to you from the whole human family.
Medicine has never before produced any single improvement of such utility.
———Thomas
Jefferson, from a letter to Edward Jenner, 1806
When Meriwether Lewis and William Clark began planning their mission
of exploring the American West, they received a special package from President
Thomas Jefferson, who had commissioned Lewis to lead the expedition.
Jefferson instructed Lewis to carry with him Jenner's smallpox vaccine
and distribute it to the Indian tribes throughout the West, many of which
had already sustained outbreaks of smallpox. Variola, the virus that causes
smallpox, had continued its expansion into the new world.
"Inform those of them with whom you may be, of it's efficacy as a preservative
from the smallpox & instruct & encourage them in the use of it," said
Jefferson.
Jefferson was hoping that the vaccine—which contained live cowpox
virus—could possibly stave off smallpox epidemics among the immunologically
naïve western tribes, whose landmass isolation had allowed them to
escape the devastating disease.
Sadly, though, the vaccine did not survive the journey west. And within
a few decades, smallpox outbreaks with mortality rates as high as 90 percent
would devastate many of the western tribes.
"The history of these things is quite interesting," says Michael B.
A. Oldstone, a professor in the Department of Neuropharmacology at The
Scripps Research Institute (TSRI), who is equally interested in viruses
themselves.
Small, Significant
The smallpox virus belongs to a family of nature's most ruthless killers—the
RNA viruses, which are among the oldest and most notorious adversaries
the human species has ever had, and which Oldstone has spent a career
studying.
They range from measles, mumps, and rubella virus to the viruses that
cause foot and mouth disease, polio, hepatitis A and C, yellow fever,
human immunodeficiency virus, and the "Spanish Flu" influenza virus, which
killed more Americans in World War I than died in combat.
In this day and age of solving genomes and mapping the expression of
thousands of genes to particular tissues, RNA viruses are somewhat of
a paradox. On the one hand, they can bring societies to their knees. Smallpox
killed more people in the 20th century than died in all the century's
wars combined.
On the other hand, RNA viruses are rather unassuming. They are like
the old VW bugs of the pathogen world—small, simple, and seemingly
able to run forever. An RNA virus can have as few as four genes. Nearly
all of them have under 10 genes. The largest RNA virus has only 12 genes.
Yet, RNA viruses are able to alter cell function and profoundly affect
the complex, eukaryotic life forms that they infect. An RNA virus with
only four genes can cripple or kill a mammal with 40,000 genes.
Take the choriomeningitis virus, for instance, one of the viruses that
Oldstone has studied for several years. Its genome is segmented into two
separate pieces of RNA that are packaged together. They are packaged inside
a compact virion, an infectious virus particle. And together, the entire
genome is only a few thousand bases—six orders of magnitude smaller
than the cells it infects—and has only four genes. Yet it can easily
persist for the lifetime of that organism.
"Everything the virus needs is in these four genes—it's amazing,"
says Oldstone, adding that, by comparison, herpes virus has 250 genes.
This leads one to ponder—as it led Oldstone to puzzle over years
ago—how, with so few genes, these RNA viruses are able to do what
they do. At the outset of his career, Oldstone decided to ask, specifically,
what are the host factors that are involved in the infection? What proteins
and other molecules in the infected organism are the viruses interacting
with? And how can we use the tens of thousands of genes in our brains
to stop those handful of viral genes? His work in this area since he began
his career at TSRI has led to recognition and many awards, including the
J. Allyn Taylor International Prize in Medicine for the study of host–virus
interactions.
The Most Contagious of Them All
One of the viruses that Oldstone has studied for a number of years,
measles, is the most contagious infectious agent known to humankind.
With an infection rate of 98 to 99 percent, measles is a highly infectious
virus that causes a maculopapular rash, fevers, diarrhea, and, one to
two times out of a thousand, death. Measles is also highly contagious,
and until the advent of mandatory vaccination programs in the United States,
there were an estimated three to four million cases annually. Some 90
percent of the U.S. population had had measles by the age of 15.
There has been a commercially available vaccine for measles in use since
1963, and, though effective, this vaccine must be kept refrigerated for
the duration of its one-year shelf life. This is problematic in tropical
climates like southern Asia and sub-Saharan Africa, which continue to
support endemic measles infection. The World Health Organization (WHO)
estimates that in 2001 there were 30 million cases of measles worldwide
and over one million deaths.
The measles virus has eight genes and gets a lot of use out of them,
infecting lymphocytes, dendritic cells, and cells of the nervous system.
The viral receptors that facilitate the entry of measles into cells are
known, and one of these receptors, called CD46, is of particular interest
to Oldstone. The measles virus has a hemagglutinin glycoprotein that binds
to a single, broad surface on one side of CD46, which is expressed on
virtually all cells in the body, and as a result, measles virus can infect
multiple organs, including the brain.
Cases of measles can be severe when the virus infects the brain, because
the body's immune response can cause massive damage to the central nervous
system, and about one out of every million cases results in a chronic,
progressive, fatal neurological disease.
However, mortality from measles is usually much higher than this because
the disease is immunosuppressive—it infects cells of the immune system—and
if "opportunistic" infectious agents like Mycobacterium tuberculosis
or Staphylococcus are present, the immunosuppression can lead to
a secondary infection, which is often fatal. Measles mortality can be
is as high as 33 percent.
Host Factors in Immunopathology
Strangely, aside from secondary infections, measles can cause a variety
of effects in a patient—called immunopathologies—and determining
the molecular causes of these is one area that interests Oldstone greatly.
Measles virus most often causes an acute infection accompanied by fevers
and characteristic rashes. Frequently, though, it also causes a subacute
infection characterized by severe brain disorders. And in some rare instances,
measles causes a chronic, progressive disease.
This is one of the great mysteries of viral infections—how they
can be so very different for different people. "How can the same virus
cause three phenotypes?" asks Oldstone.
Oldstone is able to address this question experimentally. Several years
ago, using transgenic techniques he developed the first in vivo
models that carry the measles receptors and are susceptible to the virus.
This allowed him and his laboratory to replicate a significant part of
measles infection and learn about it.
The answer to the question of divergent phenotypes seems to lie in the
field of both host genetics and viral immunopathology, in other words
in the constellation of host factors and the way in which the virus interacts
with them. These interactions determine the severity of the infection.
The great majority of symptoms that occur during an acute infection are
due to the reaction of the immune system.
"When you have an acute infection, the immune system is designed to
terminate it," says Oldstone. "Either you get immunity or you get death."
But this depends on how widespread the infection is and where it takes
place. If it takes place in the brain or the heart, the damage from the
immune reaction might be so severe that the organism cannot recover. However,
an infection that is localized in the skin might not be so severe.
Knowing what is turned on and off in the host organism and how these
molecular switches can be switched back to normal is a major goal of Oldstone's
research for another reason as well—it illuminates how viruses are
able to persist in a host for years.
The Persistence of Viral Infections
In viral infections, a battle rages in the body. Sometimes this battle
is won by the host, sometimes it is won by the virus, sometimes the host
and the virus fight each other for years in a protracted war of attrition,
and sometimes they coexist with remarkably little injury.
When a virus persists, the animal that is infected with the virus lives
a natural life but may have specific dysfunctions. If the virus lives
in the nerve cells, for instance, the learning functions of the animal
can be impaired. If the virus replicates in the immune cells, the functioning
of the immune system can be impaired.
"For a virus to persist, it has to evade the host immune system," says
Oldstone.
Viruses often distort the function of cells, but in subtle ways. For
instance, the GAP-43 gene, which is involved in cognitive function, can
be turned off by lymphocytic choriomeningitis—a virus that Oldstone
has studied for a number of years. The same virus can turn off the transcription
factor that regulates the expression of growth hormone, which results
in stunted growth.
"You don't see any distortion in the cells if you look at them under
a microscope," says Oldstone. "The brain and pituitary gland look normal."
He looks at the mechanism, asking how the virus interacts with the host
and how the host interacts with the virus. He also asks if this can be
prevented.
Interestingly, when Oldstone began studying viruses, the prevalent theory
at the time was that when viruses persisted, they did so because the host
organism mounted no host response at all to the infection. In fact, Oldstone
determined that this was not the case in the viral infections he looked
at.
"I found that the host DID make an immune response," he says, "but the
immune response was not enough to clear the infection."
And Speaking of History...
A few years ago, Oldstone wrote what might be considered an unusual
book for an academic scientist. Unusual, that is, unless one considers
the great tradition of literary scientists—Loren Eisley, Carl Sagan,
and Stephen Jay Gould to name a few—who are both accomplished scientists
and writers.
Perhaps as readers, we like to read scientist writers because their
knowledge of the field is profound, and we trust them to guide us into
the material—because we want to read about science, whether reflections
on paleontology or evolution, or the galaxies, or time and space itself.
We want to understand and we look to scientist writers who have an immediate
intimacy with the subject matter.
And fascinating subject matter it is.
Viruses, Plagues, & History (Oxford, 1998) reads like a cross
between Albert Camus and Steven Ambrose—a history wrapped in a narrative
focused on a viral immunobiologist's history of viruses interacting with
our immune system through time. His familiarity with the subjects allows
him to move seamlessly from discussing 100-year-old measles outbreaks
in Fiji to describing the immunopathology of the virus.
With chapters on smallpox, yellow fever, measles, polio, ebola, HIV,
influenza, and other subjects, Viruses has a lot of material packed
into its 230 or so pages. Still in its first edition, the book has recently
been released in paperback and has been translated into several foreign
languages, including Spanish, Polish, Chinese, Japanese, and even Hungarian.
"I've gotten invitations to speak about the book in several museums
and colleges," says Oldstone. These include the Art and Science Museum
in San Francisco and library and history departments of Ohio State, Montana,
and Augustona College.
What led him to undertake such a huge effort late in his career? Like
many of his contemporaries and many students of virology, medicine, and
public health who would follow, Oldstone was inspired by Paul de Kruif's
1926 classic Microbe Hunters. And he had other ulterior motives
as well.
"I thought it might be fun to entice undergraduates and graduates into
the field," he says. "That's why I wrote the book."
Go back to News & Views Index
|

Michael Oldstone, professor at TSRI, is also the author of a historical
narrative, Viruses, Plagues, & History.

Measles, the most contagious infectious agent known
to humankind, has afflicted many societies, as this drawing by a sixteenth-century
Aztec shows. Drawing from the Códue Florentino.
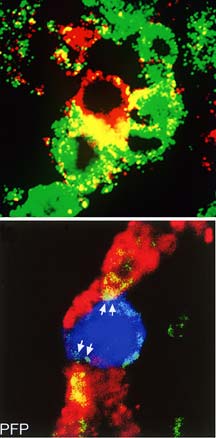
The top panel shows a single lymphocytic chorlomeningitis
virus (LCMV)-specific CD8 T killer cell engaging three virus-infected
cells in vivo in the brain. The bottom panel shows a single LCMV CD8 T
killer cell that has deposited the chemical perforin on two virus-infected
cells in the brain. Deposition of perforin is required to lyse the virus-infected
cells and thus destroy them. (Data
from studies by Dorian McGavern and M.B.A. Oldstone.)
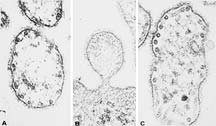
An electron micrograph showing measles particles
magnified 120,000 times. Micrography from studies by M.B.A.
Oldstone and Peter W. Lambert.
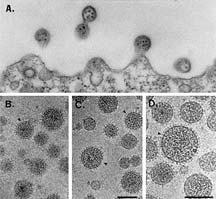
An electron micrograph showing lymphocytic choriomeningitis
virus (LCMV) virions. Micrography
from studies by M.B.A. Oldstone, Peter W. Lambert, and Michael Buchmeier.
|



